Introduction
Proteins are among the most essential macromolecules in living organisms. They perform a vast range of biological functions, including structural support, catalysis of biochemical reactions, transportation of molecules, immune defense, and cellular movement. At the molecular level, proteins are polymers made from amino acids linked together by peptide bonds.
Understanding how proteins are built, how amino acids interact, and how environmental factors such as pH influence protein structure is fundamental in fields such as biochemistry, molecular biology, medicine, and biotechnology.
This article explains the structure of proteins, the role of amino acids, peptide bond formation, protein folding, and the influence of pH on amino acid behavior.
What Are Proteins?
Proteins are long chains of amino acids connected through peptide bonds, forming complex macromolecules that perform diverse biological functions.
The terms protein and polypeptide are sometimes used interchangeably, but they have slightly different meanings:
Polypeptides: Chains of amino acids with a molecular weight less than 10,000 daltons
Proteins: Larger molecules with molecular weights greater than 10,000 daltons
Shorter chains of amino acids are known as:
Oligopeptides – chains with fewer than 20 amino acids
Peptides – very small amino acid chains
Although polypeptides form the backbone of proteins, functional proteins require proper folding into a three-dimensional structure.
Native Structure: The Functional Form of Proteins
A newly synthesized polypeptide chain does not immediately function as a protein. Instead, it must fold into a specific three-dimensional shape, known as the native structure.
The native structure is the biologically active form of a protein and is stabilized by several types of interactions between amino acid side chains.
These interactions include:
Hydrogen bonds
Hydrophobic interactions
Ionic interactions
Van der Waals forces
Disulfide bonds (in some proteins)
The side chains (R groups) of amino acids largely determine how proteins fold and maintain their structure.
If the native structure is disrupted (a process known as protein denaturation), the protein may lose its function.
Amino Acids: The Building Blocks of Proteins
Proteins are built from amino acids, small organic molecules that share a common structure.
Each amino acid contains four components attached to a central carbon atom called the alpha carbon (α-carbon):
Amino group (–NH₂)
Carboxyl group (–COOH)
Hydrogen atom
Side chain (R group)
The R group is what distinguishes one amino acid from another. It determines the chemical properties of the amino acid and influences how proteins fold and interact with other molecules.
The 20 Standard Amino Acids
The genetic code in eukaryotic organisms specifies 20 amino acids that are used in protein synthesis.
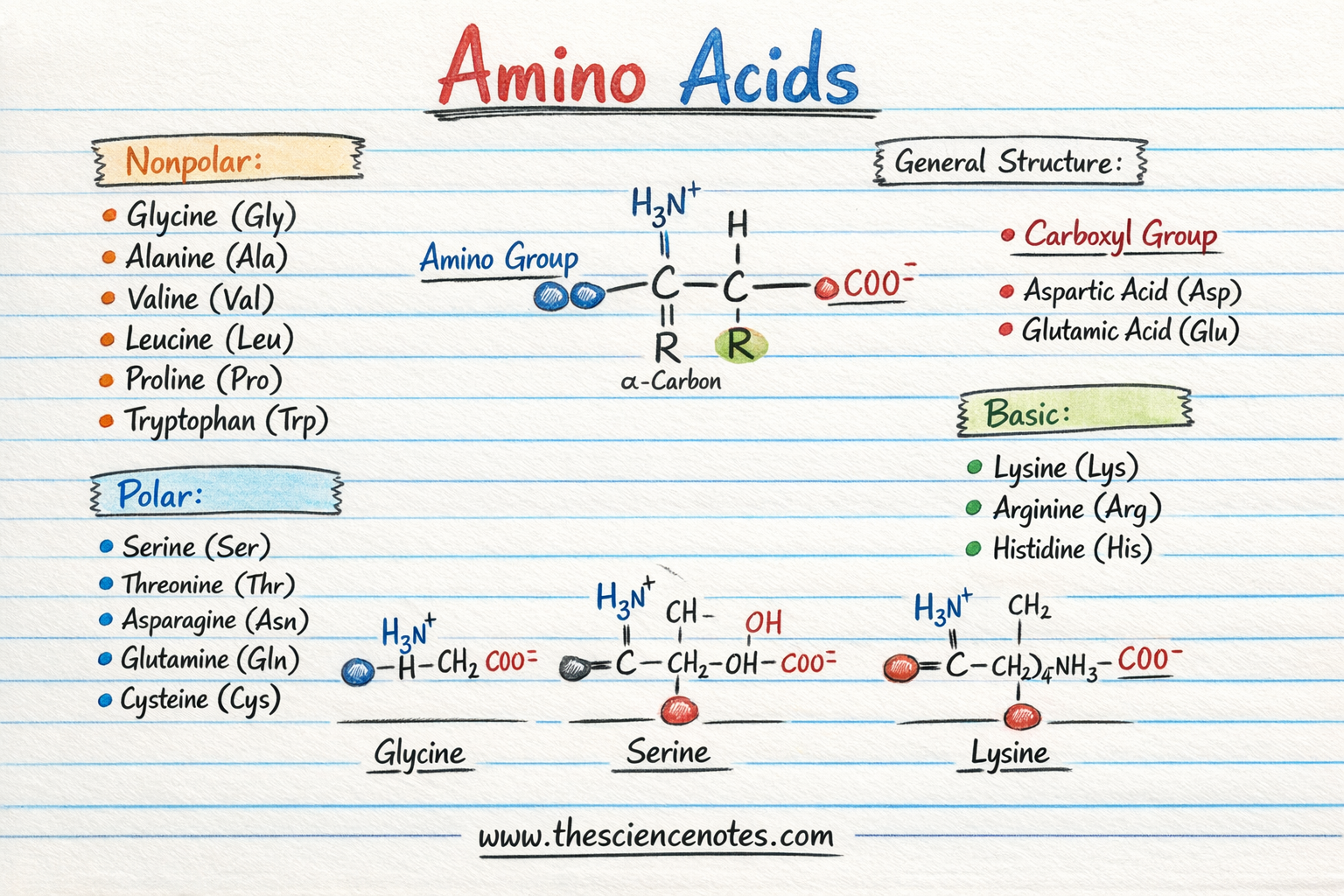
Each amino acid has:
A three-letter abbreviation
A one-letter abbreviation
Examples include:
| Amino Acid | Three-Letter Code | One-Letter Code |
|---|---|---|
| Glycine | Gly | G |
| Valine | Val | V |
| Proline | Pro | P |
| Alanine | Ala | A |
| Serine | Ser | S |
Despite sharing the same backbone structure, the diversity of R groups allows proteins to perform thousands of different biological functions.
Classification of Amino Acids Based on R Groups
Amino acids are often grouped according to the chemical properties of their side chains (R groups).
1. Acidic Amino Acids
Acidic amino acids contain a carboxyl group in their side chain.
Examples include:
Aspartic acid
Glutamic acid
These amino acids tend to donate hydrogen ions and therefore behave as acids at physiological pH.
2. Basic Amino Acids
Basic amino acids contain amine groups in their side chains.
Examples include:
Lysine
Arginine
Histidine
They tend to accept hydrogen ions and therefore act as bases.
3. Polar (Hydrophilic) Amino Acids
Some amino acids are polar but uncharged.
These amino acids:
Interact strongly with water
Form hydrogen bonds
Are usually located on the surface of proteins
Examples include:
Serine
Threonine
Asparagine
Their hydrophilic nature allows proteins to interact with the aqueous cellular environment.
4. Nonpolar (Hydrophobic) Amino Acids
Nonpolar amino acids have hydrophobic side chains that repel water.
Examples include:
Glycine
Proline
Tryptophan
Valine
Leucine
These amino acids are usually located inside the folded protein structure, away from water.
Hydrophobic interactions play a major role in protein folding and stability.
Formation of Peptide Bonds
Protein synthesis involves linking amino acids together through peptide bonds.
A peptide bond forms between:
The amino group (–NH₂) of one amino acid
The carboxyl group (–COOH) of another amino acid
During this reaction:
A molecule of water is removed
A covalent bond forms between the amino acids
Because water is released, this reaction is called a dehydration reaction (also known as a condensation reaction).
The resulting chain of amino acids is called a polypeptide.
Directionality of Polypeptide Chains
Polypeptide chains have directionality, meaning they have two distinct ends.
N-terminus
The N-terminus contains the free amino group (–NH₃⁺).
This is the starting point of the polypeptide chain.
C-terminus
The C-terminus contains the free carboxyl group (–COO⁻).
This marks the end of the protein chain.
Protein sequences are always written and read from:
N-terminus → C-terminus
This direction is important during protein synthesis and molecular biology studies.
The Polypeptide Backbone
When amino acids join together, they form a repeating pattern of atoms called the polypeptide backbone.
This backbone consists of repeating units of:
Nitrogen
Carbon
Carbon
The R groups extend outward from the backbone, allowing them to interact with each other and the surrounding environment.
These interactions determine the final folded structure of the protein.
Role of pH in Amino Acid Behavior
Amino acids contain both acidic and basic functional groups, meaning they can act as either acids or bases depending on the surrounding pH.
This property is known as amphoteric behavior.
Low pH (Acidic Environment)
At low pH levels (around pH 2):
Amino group becomes protonated (–NH₃⁺)
Carboxyl group remains –COOH
Under these conditions, amino acids behave more like bases.
High pH (Alkaline Environment)
At high pH levels (around pH 13):
Amino group becomes –NH₂
Carboxyl group becomes –COO⁻
In this environment, amino acids behave more like acids.
Physiological pH (Neutral Environment)
In most biological systems, the pH is approximately 7.4.
At this pH:
Amino group is protonated (–NH₃⁺)
Carboxyl group is deprotonated (–COO⁻)
This creates a molecule called a zwitterion.
A zwitterion carries:
One positive charge
One negative charge
This property allows amino acids to form hydrogen bonds and ionic interactions, which are critical for protein folding and stability.
Diversity and Functions of Proteins
Proteins exhibit enormous diversity in structure, composition, and function. This diversity arises from the different combinations of 20 amino acids arranged in unique sequences.
Some major biological functions of proteins include:
Structural Proteins
Structural proteins provide support and strength to tissues.
Example:
Collagen – found in connective tissues such as skin, bones, and cartilage
Contractile Proteins
Contractile proteins allow muscle contraction and cellular movement.
Examples include:
Actin
Myosin
These proteins interact to generate muscle movement.
Enzymes
Many proteins function as enzymes, which act as biological catalysts.
Enzymes:
Speed up chemical reactions
Lower activation energy
Are highly specific to their substrates
Examples include:
DNA polymerase
Amylase
Proteases
Transport Proteins
Transport proteins help move molecules across cell membranes.
Examples include:
Ion channels
Carrier proteins
These proteins regulate the movement of substances such as:
Glucose
Ions
Water
Immune Defense Proteins
Proteins also protect organisms against pathogens and foreign substances.
Example:
Antibodies (immunoglobulins)
Antibodies recognize and bind to specific antigens, helping the immune system eliminate infections.
Why Protein Structure Determines Function
One of the most important principles in biology is:
Protein structure determines protein function.
Even a small change in amino acid sequence can affect protein folding and alter its function.
For example:
Mutations in proteins can cause genetic diseases
Misfolded proteins are associated with disorders such as Alzheimer’s disease and prion diseases
Therefore, understanding protein structure is critical in:
Drug design
Molecular biology
Biotechnology
Medicine
Conclusion
Proteins are complex biological macromolecules composed of amino acids linked by peptide bonds. The sequence of amino acids determines how a protein folds into its native three-dimensional structure, which ultimately defines its biological function.
The 20 amino acids encoded by the genetic code provide immense structural and functional diversity. Their side chains, chemical properties, and interactions with water and other molecules drive protein folding and stability.
Environmental factors such as pH influence amino acid charge states and contribute to the formation of zwitterions, hydrogen bonds, and other interactions that shape protein structure.
From structural support and muscle movement to enzyme catalysis and immune defense, proteins are indispensable to life. A deeper understanding of protein structure and amino acid chemistry continues to drive advances in biochemistry, medicine, and biotechnology.
LEARN MORE: Proteins and Amino Acids: Structure, Function, and Biological Importance – The Science Notes