B-cell maturation antigen (BCMA), also known as TNFRSF17 (in BCMA signaling), is a critical survival receptor expressed predominantly on plasma cells and malignant plasma cells in multiple myeloma. As a member of the tumor necrosis factor receptor (TNFR) superfamily, BCMA plays an essential role in regulating plasma cell proliferation, differentiation, and survival through interaction with its ligands BAFF (B-cell activating factor) and APRIL (a proliferation-inducing ligand). Activation of BCMA triggers downstream signaling pathways including NF-κB, PI3K–AKT, and MAPK, which promote tumor growth and resistance to apoptosis. Due to its restricted expression pattern and central role in plasma cell biology, BCMA has emerged as one of the most promising therapeutic targets in multiple myeloma. This review explores the molecular structure of BCMA, its physiological role in B-cell development, signaling mechanisms, and the therapeutic strategies designed to target BCMA in cancer immunotherapy.
Introduction
Multiple myeloma is a hematologic malignancy characterized by the uncontrolled proliferation of plasma cells within the bone marrow. These malignant plasma cells produce abnormal immunoglobulins and disrupt normal hematopoiesis.
Recent advances in cancer immunotherapy have identified BCMA (B-cell maturation antigen) as a key molecular target in multiple myeloma.
BCMA is uniquely suited for targeted therapy because it exhibits:
High expression on malignant plasma cells
Minimal expression in normal tissues
A critical role in plasma cell survival signaling
These characteristics have made BCMA the focus of several innovative therapies including:
Bispecific antibodies
CAR-T cell therapy
Antibody-drug conjugates
Understanding the BCMA signaling pathway is essential for developing effective treatments and overcoming therapeutic resistance.
Molecular Structure of BCMA
BCMA (TNFRSF17) is a type III transmembrane receptor belonging to the tumor necrosis factor receptor superfamily.
The receptor contains several structural components:
Extracellular domain
This domain binds the ligands BAFF and APRIL.
Transmembrane region
Anchors the receptor within the plasma membrane.
Intracellular cytoplasmic domain
Interacts with adaptor proteins that initiate downstream signaling cascades.
Unlike many other TNF receptors, BCMA has a relatively short cytoplasmic tail but still efficiently recruits signaling molecules that regulate plasma cell survival.
Expression Pattern of BCMA
BCMA expression is tightly regulated during B-cell development.
BCMA is primarily expressed on:
Mature plasma cells
Long-lived plasma cells
Malignant plasma cells in multiple myeloma
Importantly, BCMA is not significantly expressed on hematopoietic stem cells or most non-immune tissues, which minimizes off-target toxicity when used as a therapeutic target.
This restricted expression pattern makes BCMA particularly suitable for targeted immunotherapy.
BCMA Ligands: BAFF and APRIL
BCMA signaling is activated by two primary ligands:
BAFF (B-cell Activating Factor)
BAFF is a cytokine belonging to the TNF family and is produced by:
Monocytes
Dendritic cells
Stromal cells
BAFF regulates B-cell maturation and survival.
APRIL (A Proliferation-Inducing Ligand)
APRIL is another TNF family cytokine that binds BCMA with higher affinity than BAFF.
APRIL is produced by:
Myeloid cells
Bone marrow stromal cells
Osteoclasts
APRIL–BCMA interaction is particularly important in multiple myeloma progression, as it promotes tumor cell survival within the bone marrow microenvironment.
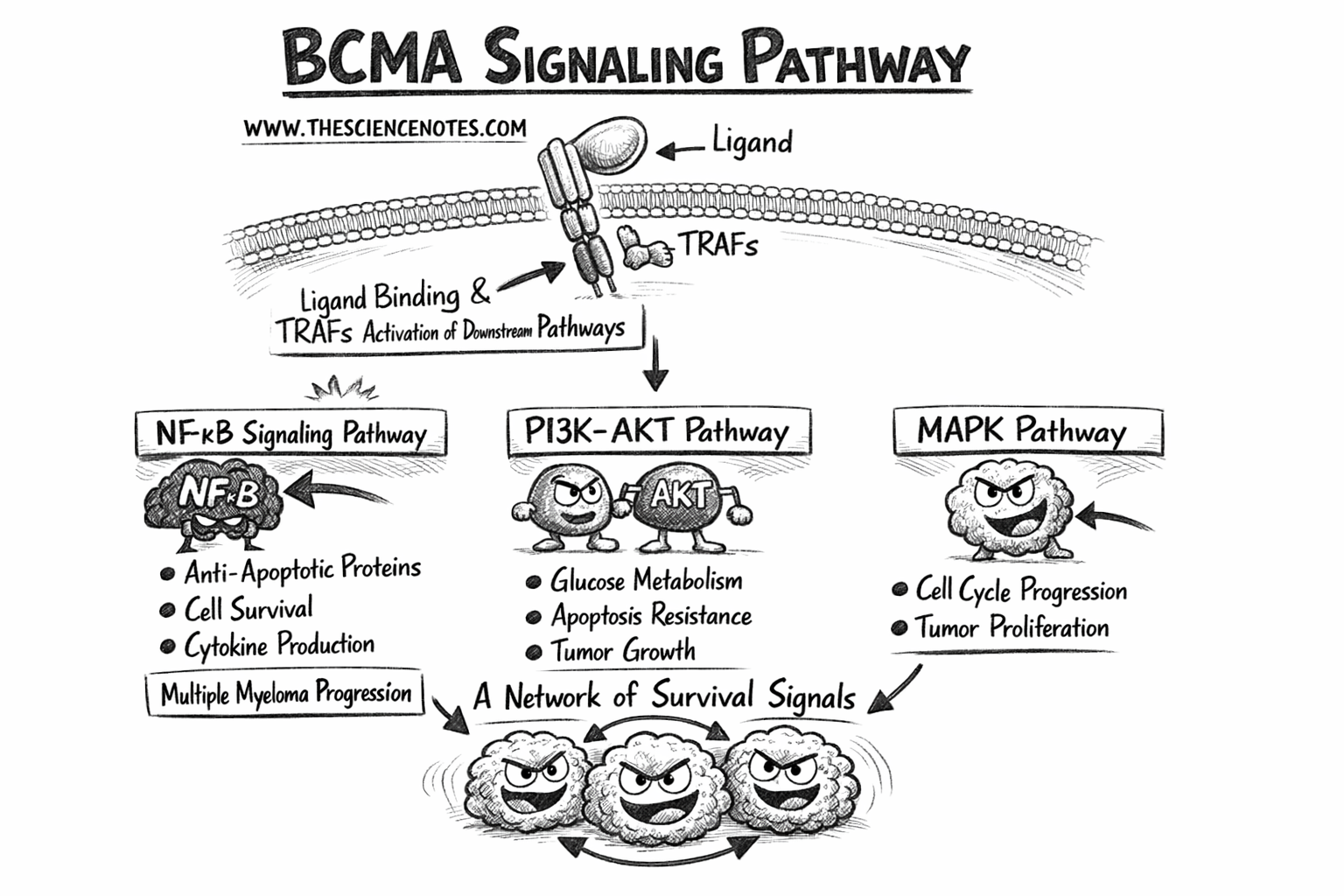
BCMA Signaling Pathway
Upon ligand binding, BCMA recruits adaptor proteins that initiate several intracellular signaling pathways.
Key signaling components include TNF receptor-associated factors (TRAFs).
These adaptor proteins activate multiple downstream pathways.
NF-κB Signaling Pathway
One of the most important pathways activated by BCMA is NF-κB signaling.
Activation of NF-κB leads to:
Increased expression of anti-apoptotic proteins
Enhanced cell survival
Increased cytokine production
NF-κB signaling plays a central role in multiple myeloma pathogenesis.
PI3K–AKT Signaling Pathway
The PI3K–AKT pathway regulates cellular metabolism, proliferation, and survival.
Activation of this pathway results in:
Increased glucose metabolism
Resistance to apoptosis
Enhanced tumor cell proliferation
This pathway contributes to therapy resistance in multiple myeloma.
MAPK Signaling Cascade
The MAPK pathway regulates cell growth and differentiation.
Activation leads to:
ERK phosphorylation
Increased cell cycle progression
Enhanced tumor proliferation
Together, these signaling pathways create a powerful survival network for malignant plasma cells.
Role of BCMA in Multiple Myeloma Pathogenesis
BCMA signaling contributes to multiple aspects of myeloma biology.
Tumor Cell Survival
Activation of BCMA promotes anti-apoptotic signaling, allowing tumor cells to evade programmed cell death.
Interaction With the Bone Marrow Microenvironment
Myeloma cells interact with stromal cells, osteoclasts, and immune cells in the bone marrow niche.
These cells produce APRIL and BAFF, which continuously activate BCMA signaling.
Immune Evasion
BCMA signaling may contribute to immune evasion by altering cytokine production and immune cell recruitment.
Soluble BCMA and Disease Monitoring
BCMA can be cleaved from the cell surface by γ-secretase, producing soluble BCMA (sBCMA).
Elevated sBCMA levels are associated with:
Higher tumor burden
Disease progression
Poor prognosis
sBCMA is increasingly used as a biomarker for monitoring treatment response in multiple myeloma.
Therapeutic Targeting of BCMA
Because of its critical biological role, BCMA has become one of the most important targets in modern multiple myeloma therapy.
Three major therapeutic approaches target BCMA.
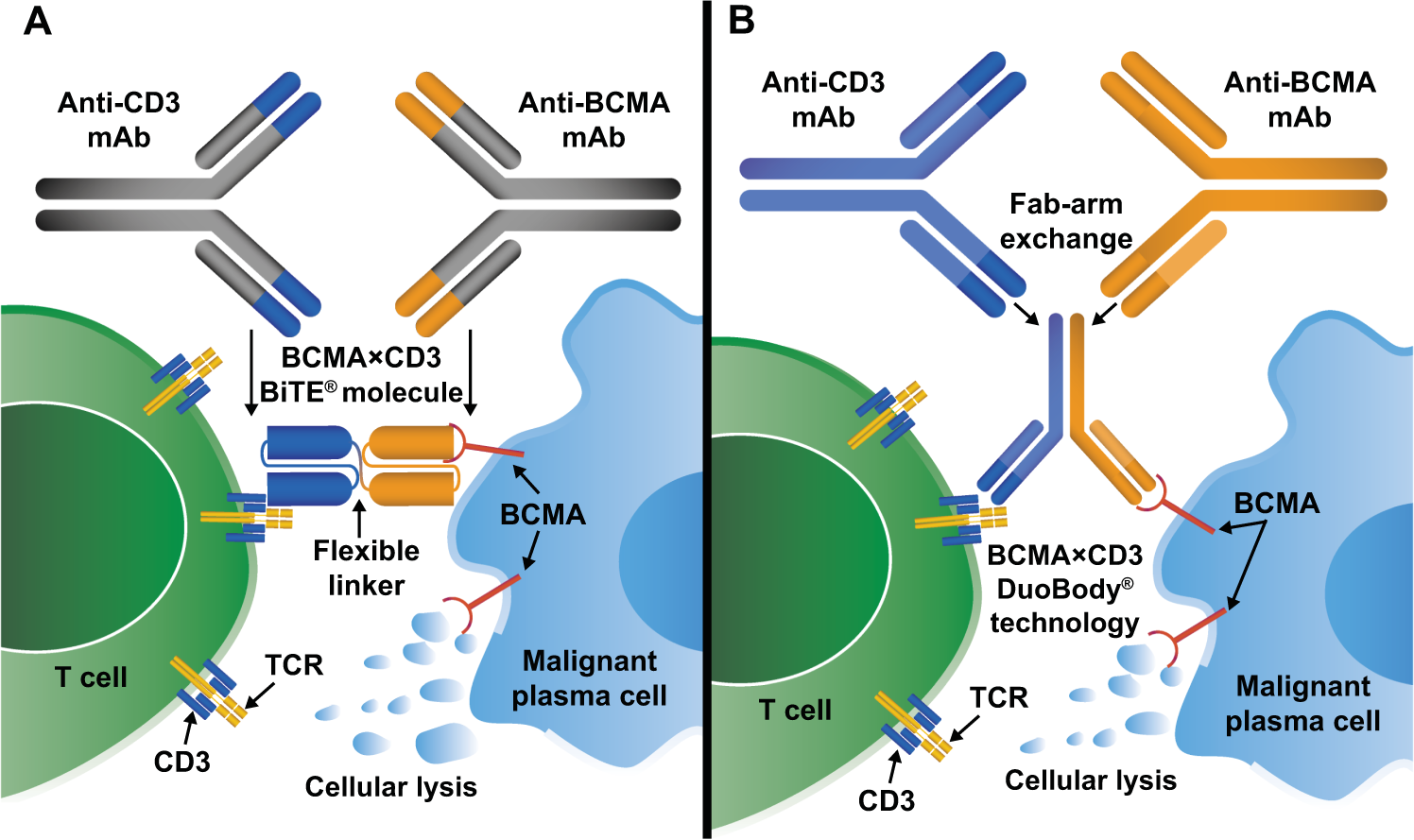
Bispecific Antibodies
Bispecific antibodies simultaneously bind BCMA on tumor cells and CD3 on T cells.
This interaction redirects cytotoxic T lymphocytes toward malignant plasma cells.
Examples include:
Teclistamab
Elranatamab
These therapies represent an important off-the-shelf immunotherapy option.
CAR-T Cell Therapy
CAR-T therapy involves genetically modifying a patient’s T cells to recognize BCMA.
Once infused, these engineered T cells can:
Recognize BCMA-expressing cells
Proliferate in vivo
Destroy tumor cells
Examples include:
Idecabtagene vicleucel
Ciltacabtagene autoleucel
Antibody-Drug Conjugates
Antibody-drug conjugates deliver cytotoxic drugs directly to BCMA-expressing tumor cells.
The antibody binds BCMA, and the internalized drug payload induces tumor cell death.
Example:
Belantamab mafodotin
Resistance Mechanisms to BCMA-Targeted Therapies
Despite impressive clinical responses, resistance may develop.
Major mechanisms include:
BCMA Antigen Loss
Tumor cells may reduce or eliminate BCMA expression.
T-Cell Exhaustion
Chronic immune activation can lead to dysfunctional T cells with reduced cytotoxic capacity.
Immunosuppressive Microenvironment
Bone marrow stromal cells and regulatory immune cells can inhibit anti-tumor immune responses.
Future Directions
Emerging research aims to enhance BCMA-targeted therapies through:
Dual-antigen targeting antibodies
Combination immunotherapies
Improved CAR-T cell engineering
Personalized treatment strategies
These innovations may further improve outcomes for patients with multiple myeloma.
Conclusion
BCMA plays a central role in plasma cell survival and multiple myeloma progression through activation of key signaling pathways including NF-κB, PI3K–AKT, and MAPK. Its restricted expression pattern and biological importance make BCMA an ideal therapeutic target. Recent advances in immunotherapy—including bispecific antibodies, CAR-T cell therapy, and antibody-drug conjugates—have transformed the treatment landscape for patients with relapsed or refractory multiple myeloma. Continued research into BCMA biology and signaling will be critical for overcoming resistance mechanisms and improving long-term clinical outcomes.
Frequently Asked Questions (FAQ)
Q1. What does BCMA stand for?
BCMA stands for B-cell maturation antigen, a receptor expressed primarily on plasma cells.
Q2. Why is BCMA important in multiple myeloma?
BCMA promotes plasma cell survival and proliferation through activation of NF-κB and PI3K–AKT signaling pathways.
Q3. What therapies target BCMA?
Major BCMA therapies include bispecific antibodies, CAR-T cell therapy, and antibody-drug conjugates.
Q4. What is soluble BCMA?
Soluble BCMA is a circulating form of BCMA released from the cell surface and can be used as a biomarker for disease monitoring.
Q5. Why is BCMA considered an ideal therapeutic target?
BCMA is highly expressed on malignant plasma cells but minimally expressed in normal tissues, reducing off-target toxicity.