Author: Alisha G C
Abstract
B-cell maturation antigen (BCMA) has emerged as one of the most promising therapeutic targets in multiple myeloma due to its selective expression on plasma cells and critical role in tumor survival signaling. Several innovative immunotherapies targeting BCMA have recently transformed the treatment landscape of relapsed or refractory multiple myeloma (RRMM). These include bispecific antibodies such as Teclistamab, chimeric antigen receptor T-cell (CAR-T) therapies including Idecabtagene vicleucel and Ciltacabtagene autoleucel, and antibody-drug conjugates such as Belantamab mafodotin. Each therapeutic modality uses a distinct mechanism to eliminate malignant plasma cells while overcoming resistance to conventional therapies such as proteasome inhibitors, immunomodulatory drugs, and anti-CD38 antibodies. This review provides a comprehensive comparison of BCMA-targeted therapies, focusing on their molecular design, mechanisms of action, clinical efficacy, safety profiles, and emerging resistance mechanisms.
Introduction
Multiple myeloma is a malignant plasma cell disorder characterized by clonal expansion of antibody-producing plasma cells within the bone marrow. Despite advances in treatment using proteasome inhibitors, immunomodulatory drugs, and monoclonal antibodies, most patients eventually develop relapsed or refractory disease.
Recent progress in cancer immunotherapy has identified BCMA (B-cell maturation antigen) as a highly effective therapeutic target.
BCMA is expressed almost exclusively on:
Plasma cells
Malignant myeloma cells
Importantly, it plays a crucial role in plasma cell survival by interacting with its ligands BAFF and APRIL, which activate intracellular pathways such as:
NF-κB signaling
PI3K–AKT pathway
MAP kinase signaling
Because of its restricted expression and biological importance, BCMA has become the focus of several next-generation immunotherapies.
Three major BCMA-targeted therapeutic classes have emerged:
Bispecific antibodies
CAR-T cell therapies
Antibody-drug conjugates
Each approach uses a unique strategy to destroy malignant plasma cells.
Bispecific Antibodies Targeting BCMA
In BCMA-targeted therapy, these antibodies bind:
BCMA on myeloma cells
CD3 on T cells
This dual binding brings cytotoxic T lymphocytes into direct contact with tumor cells and triggers immune-mediated killing.
Teclistamab
Teclistamab is a BCMA × CD3 bispecific IgG4 antibody designed to redirect T cells toward malignant plasma cells.
Once both targets are engaged:
T-cell receptor clustering occurs
T cells become activated
Cytotoxic granules containing perforin and granzyme B are released
Tumor cells undergo apoptosis
Unlike CAR-T therapy, Teclistamab uses the patient’s existing T cells without genetic modification.
Advantages of Bispecific Antibodies
Off-the-shelf therapy
No complex manufacturing
Repeat dosing possible
Rapid treatment initiation
Limitations
Cytokine release syndrome
Risk of infection due to immune suppression
Possible antigen escape
CAR-T Cell Therapy Targeting BCMA
Chimeric antigen receptor T-cell (CAR-T) therapy represents one of the most advanced forms of personalized cancer immunotherapy.
In this approach:
T cells are collected from the patient.
These cells are genetically engineered to express a CAR receptor targeting BCMA.
The modified cells are expanded in the laboratory.
CAR-T cells are infused back into the patient.
Once infused, CAR-T cells recognize BCMA-expressing tumor cells and trigger strong cytotoxic immune responses.
Two BCMA CAR-T therapies have shown remarkable clinical success.
Idecabtagene Vicleucel (Abecma)
This CAR-T therapy demonstrated high response rates in heavily pretreated patients.
Key clinical findings include:
Overall response rate ~73%
Median progression-free survival ~8–9 months
Ciltacabtagene Autoleucel (Carvykti)
Ciltacabtagene autoleucel has demonstrated even higher response rates.
Clinical trials reported:
Overall response rate >95%
Deep and durable responses in many patients
Advantages of CAR-T Therapy
Extremely high response rates
Potential for long-term remission
Single infusion therapy
Limitations
Complex manufacturing process
Long production time
High treatment cost
Risk of severe cytokine release syndrome and neurotoxicity
Antibody-Drug Conjugates Targeting BCMA
Antibody-drug conjugates (ADCs) combine the specificity of monoclonal antibodies with the cytotoxic potency of chemotherapy.
These molecules consist of three components:
Monoclonal antibody targeting BCMA
Cytotoxic drug payload
Chemical linker
After binding BCMA:
The ADC is internalized into the tumor cell.
The linker is cleaved.
The cytotoxic payload is released.
Tumor cell death occurs.
Belantamab Mafodotin
Belantamab mafodotin was the first BCMA-targeted ADC approved for multiple myeloma.
Its cytotoxic payload monomethyl auristatin F (MMAF) disrupts microtubule formation and induces apoptosis.
Advantages of Antibody-Drug Conjugates
Direct tumor killing
No reliance on immune activation
Simpler administration compared with CAR-T therapy
Limitations
Ocular toxicity (keratopathy)
Lower response rates compared with CAR-T therapies
Potential drug resistance
Comparison of BCMA-Targeted Therapies
| Feature | Bispecific Antibodies (Teclistamab) | CAR-T Therapy | Antibody-Drug Conjugates |
|---|---|---|---|
| Mechanism | T-cell redirection | Engineered T cells | Cytotoxic drug delivery |
| Manufacturing | Off-the-shelf | Personalized | Off-the-shelf |
| Administration | Repeated dosing | Single infusion | Repeated dosing |
| Response rates | ~60–65% | 70–95% | ~30–35% |
| Toxicity | CRS, infections | CRS, neurotoxicity | Ocular toxicity |
Resistance Mechanisms in BCMA-Targeted Therapies
Despite impressive responses, resistance remains a major challenge.
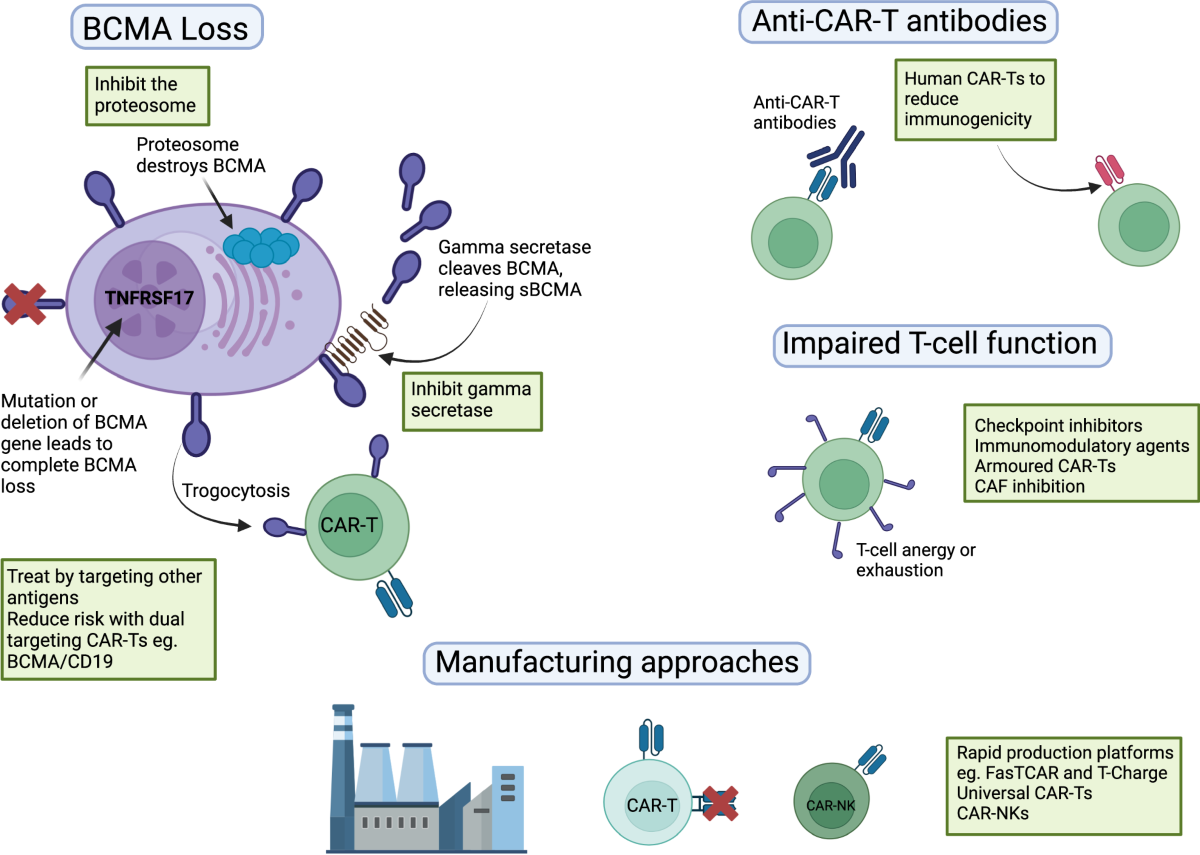
BCMA Antigen Loss
Tumor cells may reduce or eliminate BCMA expression, preventing immune recognition.
Mechanisms include:
Genetic mutations
Alternative splicing
Shedding of soluble BCMA
T-Cell Dysfunction
Chronic immune activation can lead to T-cell exhaustion, characterized by:
Increased PD-1 expression
Reduced cytokine production
Impaired cytotoxicity
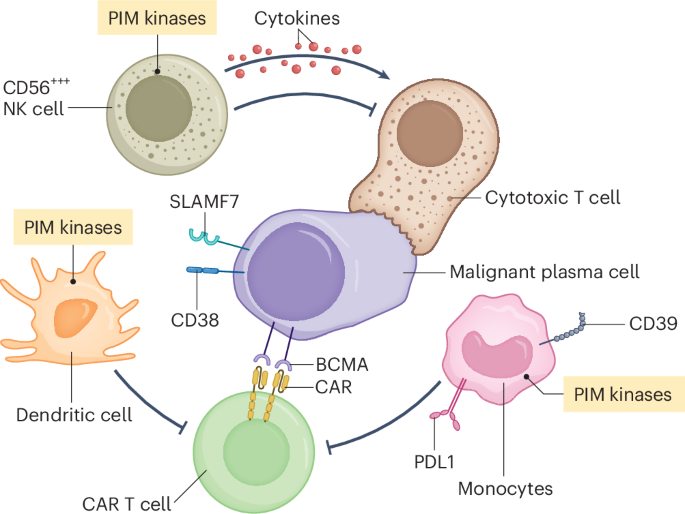
Immunosuppressive Bone Marrow Microenvironment
The myeloma bone marrow niche contains suppressive immune cells including:
Regulatory T cells
Myeloid-derived suppressor cells
Tumor-associated macrophages
These cells produce inhibitory cytokines that dampen anti-tumor immune responses.
Emerging BCMA-Targeted Strategies
Next-generation therapies are being developed to improve treatment durability.
Dual-Target Bispecific Antibodies
New agents target multiple plasma cell antigens simultaneously, such as:
BCMA + GPRC5D
BCMA + FcRH5
This strategy reduces antigen escape.
Combination Immunotherapy
Combining BCMA therapies with:
Checkpoint inhibitors
Immunomodulatory drugs
Cytokine therapies
may improve T-cell function and persistence.
Future Directions
The future of multiple myeloma treatment will likely involve combination immunotherapy approaches integrating:
Bispecific antibodies
CAR-T cell therapy
Antibody-drug conjugates
Immune checkpoint inhibitors
Advances in immune engineering and molecular profiling will enable personalized treatment strategies for patients with relapsed or refractory disease.
Conclusion
BCMA-targeted therapies have revolutionized the treatment of multiple myeloma by providing highly effective immunotherapeutic options for patients with refractory disease. Bispecific antibodies such as Teclistamab enable rapid off-the-shelf immune redirection, while CAR-T cell therapies offer deep and durable responses through engineered cellular immunity. Antibody-drug conjugates provide a complementary approach by delivering potent cytotoxic agents directly to tumor cells. Continued research aimed at overcoming resistance mechanisms and optimizing combination strategies will be critical for improving long-term outcomes and achieving durable remissions in multiple myeloma.
Frequently Asked Questions (FAQ)
Q1. What is BCMA and why is it important in multiple myeloma?
BCMA is a plasma cell surface receptor that regulates survival signaling and is highly expressed on malignant myeloma cells, making it an ideal therapeutic target.
Q2. What are the three major BCMA-targeted therapies?
The main classes include bispecific antibodies, CAR-T cell therapy, and antibody-drug conjugates.
Q3. Which BCMA therapy is most effective?
CAR-T therapies currently show the highest response rates, but bispecific antibodies offer faster and more accessible treatment.
Q4. What are the major side effects of BCMA immunotherapy?
Common toxicities include cytokine release syndrome, infections, neurotoxicity, and ocular toxicity depending on the therapy type.
Q5. Can patients receive multiple BCMA therapies?
Yes, sequential or combination therapies are being explored to overcome resistance and improve outcomes.