Red blood cells (RBCs), scientifically known as erythrocytes, are the vital transport vehicles of the human cardiovascular system. Their primary mission is to deliver oxygen to every tissue in the body, ensuring cellular respiration and survival. However, these cells have a rigorous “shelf life” of only 120 days. To sustain life, the body must constantly engage in a sophisticated biological manufacturing process called erythropoiesis.
In this comprehensive guide, we explore the intricate mechanics of how red blood cells are formed, the role of hormones like erythropoietin (EPO), the unique structure of hemoglobin, and the medical disorders that arise when this process falters.
What is Erythropoiesis?
Erythropoiesis is the specific physiological process by which red blood cells are generated. In a healthy adult, this occurs primarily within the red bone marrow. This system is designed to be highly adaptive; it ensures all tissues are amply supplied with oxygen while preventing an over-proliferation of cells that could thicken the blood and impair flow.
The rate of production is largely determined by tissue oxygen levels. When oxygen levels drop—a condition known as hypoxia—the body immediately signals the bone marrow to ramp up production to maintain homeostasis.
The Role of Erythropoietin (EPO): The Master Regulator
The initiation of erythropoiesis is governed by a glycoprotein hormone called Erythropoietin (EPO).
Sensing Oxygen Shortages
While the fetal liver initially produces EPO, post-birth production shifts to the interstitial cells of the kidney. These cells act as biological sensors. When they detect a drop in physiological oxygen levels—due to injury, blood loss, or environmental factors like high altitude—they trigger a molecular response.
The HIF-1ɑ Mechanism
Low oxygen levels prevent the degradation of a transcription factor known as HIF-1ɑ. As HIF-1ɑ accumulates, it transcribes the EPO gene, causing more of the hormone to be released into the bloodstream. Once released, EPO travels to the bone marrow and binds to erythropoietin receptors (EpoR) on erythroid progenitor cells, setting the maturation process in motion.
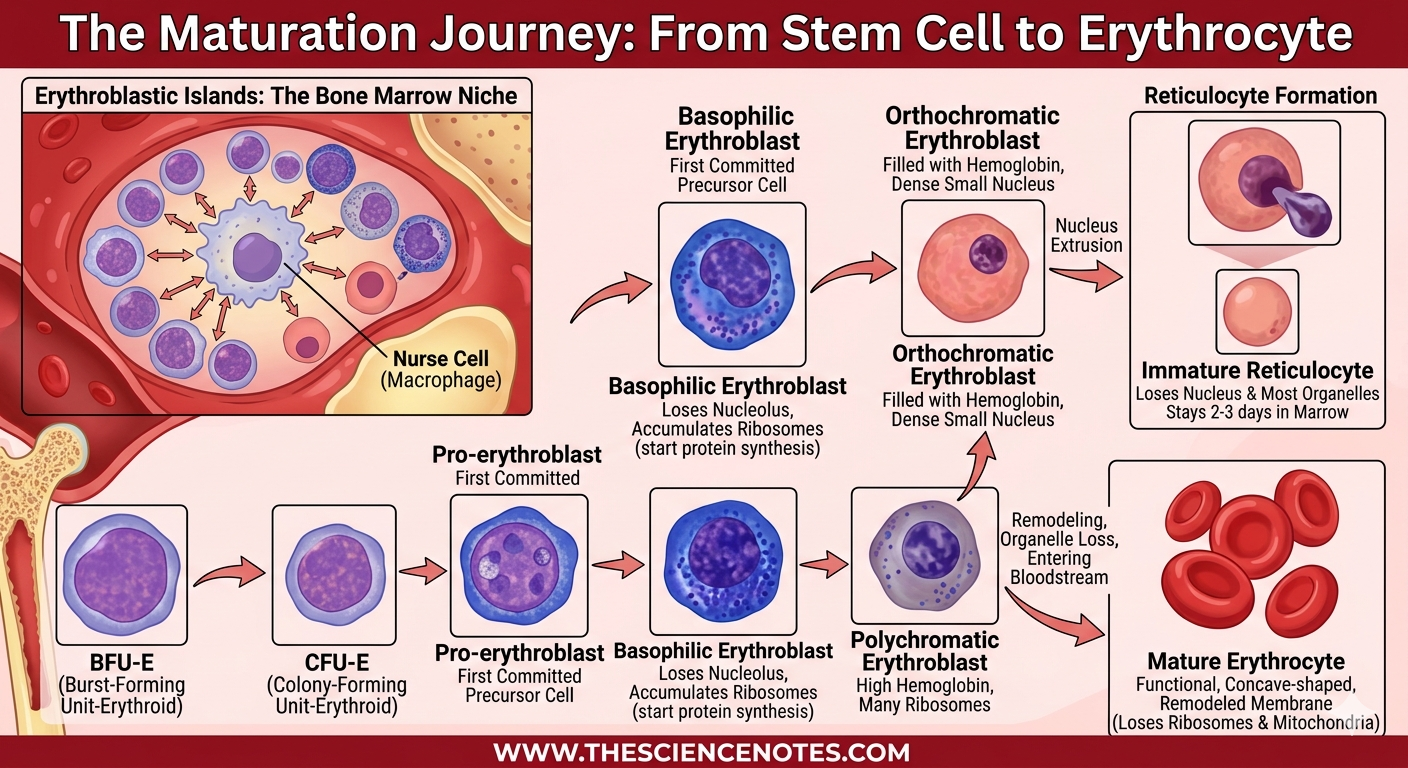
The Maturation Journey: From Stem Cell to Erythrocyte
Red blood cells undergo a series of complex differentiation and maturation stages within “erythroblastic islands”—specialized niches in the bone marrow where progenitors interact with a central macrophage, often referred to as a “nurse cell.”
Stages of RBC Development:
Progenitor Stages: It begins with the Burst-Forming Unit-Erythroid (BFU-E), which differentiates into the Colony-Forming Unit-Erythroid (CFU-E).
Pro-erythroblast: The first committed precursor cell.
Basophilic Erythroblast: At this stage, the cell loses its nucleolus and begins to accumulate ribosomes to start protein synthesis.
Polychromatic Erythroblast: Characterized by a high concentration of hemoglobin and numerous ribosomes.
Orthochromatic Erythroblast: The cell becomes filled with hemoglobin, and its nucleus becomes small and dense.
Reticulocyte Formation: The cell expels its nucleus and loses most organelles. These immature reticulocytes stay in the marrow for 2–3 days before entering the bloodstream.
Mature Erythrocyte: After final membrane remodeling in the blood, where they lose remaining ribosomes and mitochondria, they become the functional, concave-shaped cells we recognize.
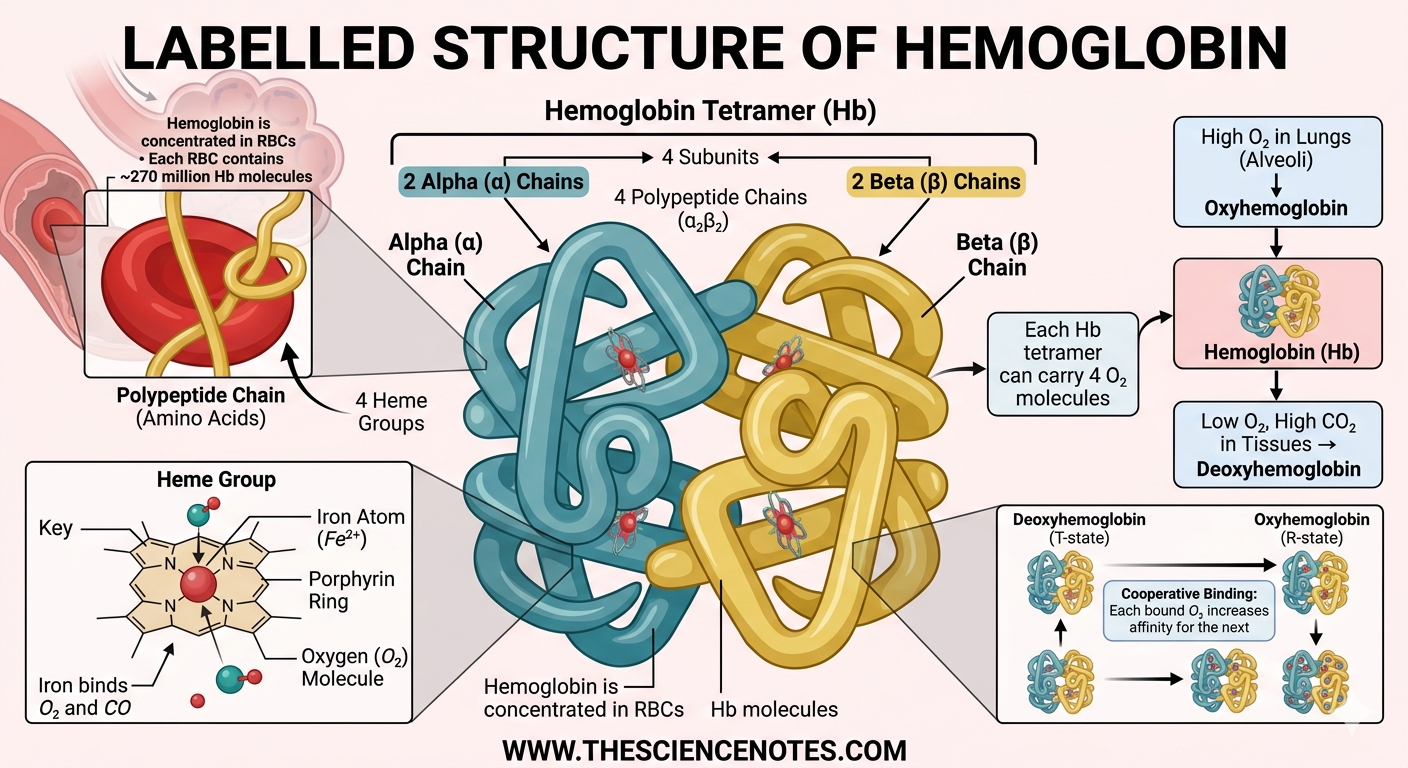
Hemoglobin: The Oxygen-Binding Protein
A single microliter of blood contains between 4.2 and 6 million erythrocytes. Nearly 97% of the protein within these cells is hemoglobin, a tetrameric globular protein designed for gas exchange.
Structure and Function
Hemoglobin is composed of four subunits: two alpha chains and two beta chains. Each chain contains a heme group with an iron atom at its core.
Oxyhemoglobin: When all four heme groups bind to oxygen, the molecule is saturated. This gives arterial blood its bright red hue.
Deoxyhemoglobin: After releasing oxygen to the tissues, hemoglobin becomes deoxyhemoglobin, resulting in the darker red appearance of venous blood.
Carbon Dioxide Transport: Hemoglobin also forms carbaminohemoglobin, transporting about 20% of the body’s $CO_2$ back to the lungs.
Nitric Oxide Binding: Hemoglobin can bind nitric oxide, which helps induce vasodilation, widening blood vessels to improve local blood flow.
The Ingenious Design of the Red Blood Cell
The mature erythrocyte is a masterpiece of evolutionary engineering. Its unique physical characteristics are specifically optimized for its environment:
Biconcave Shape: This flattened disc shape provides 30% more surface area relative to volume than a sphere. This facilitates a swift exchange of gases.
Flexibility and Spectrin: The plasma membrane is associated with a protein meshwork called spectrin. This allows the cell to twist, turn, and become cup-shaped to navigate narrow capillaries without breaking.
Anaerobic Metabolism: Because erythrocytes lack mitochondria, they do not consume the oxygen they carry. They generate ATP through anaerobic processes, making them highly efficient transporters.
Hemoglobin: Structure, Confirmation, Binding and Transportation of Oxygen
Common Disorders of the Erythrocytes
When the delicate balance of erythropoiesis is disturbed, it leads to significant physiological disorders. These are generally categorized into Anemia and Polycythemia.
Anemic Conditions (Low Oxygen Capacity)
Anemia occurs when the blood has a reduced capacity to carry oxygen due to loss, lower production, or destruction of RBCs.
Hemorrhagic Anemia: Resulting from acute or chronic blood loss from wounds or ulcers.
Iron Deficiency Anemia: Caused by a lack of iron needed for hemoglobin synthesis.
Pernicious Anemia: Linked to stomach mucosa atrophy, which reduces Vitamin B12 absorption and leads to the production of short-lived macrocytes.
Sickle Cell Disease: A genetic disorder where abnormal hemoglobin causes cells to “sickle,” leading to painful blockages and premature cell death.
Thalassemia: A genetic condition resulting in abnormal hemoglobin production and fragile RBCs.
Polycythemic Conditions (Overproduction)
Polycythemia involves an overproduction of red blood cells, which increases blood viscosity and can lead to clotting or stroke.
Primary Polycythemia (Polycythemia Vera): Often due to genetic mutations in the bone marrow.
Secondary Polycythemia: A result of conditions like chronic hypoxia (high altitude or smoking) that force the body to overproduce EPO.
Conclusion: Maintaining Blood Health
Healthy erythropoiesis is a cornerstone of overall wellness. Nutritional factors, specifically Iron, Vitamin B9 (Folate), and Vitamin B12, are essential for DNA synthesis and hemoglobin production. By understanding this 120-day cycle of renewal, we can better appreciate the complex cardiovascular mechanisms that keep our bodies oxygenated and thriving.
Frequently Asked Questions About Erythropoiesis and Red Blood Cells
Below are some of the most common questions regarding how our bodies produce, manage, and utilize red blood cells (RBCs).
1. What is the main purpose of erythropoiesis?
The primary goal of erythropoiesis is to maintain a stable number of erythrocytes (red blood cells) in the bloodstream. This ensures that all body tissues receive an adequate and continuous supply of oxygen for cellular metabolism. It also replaces old or damaged cells, as RBCs only live for about 120 days.
2. How does the body know when to produce more red blood cells?
The process is regulated by a feedback loop involving the kidneys. Specialized cells in the kidneys sense a drop in oxygen levels (hypoxia). In response, they release the hormone erythropoietin (EPO), which travels to the bone marrow to stimulate the production of new RBCs from hematopoietic stem cells.
3. Why do mature red blood cells lack a nucleus?
Mature erythrocytes expel their nuclei and other organelles (like mitochondria) during the final stages of development. This serves two main purposes:
Increased Space: It creates more room for hemoglobin, which makes up about 97% of the cell’s internal protein.
Efficiency: By lacking mitochondria, RBCs do not consume the oxygen they are transporting, ensuring the maximum amount reaches the tissues.
4. What are “reticulocytes” and why are they important?
Reticulocytes are immature red blood cells that have just lost their nuclei but still contain some residual ribosomes and organelles. They typically circulate in the blood for about 24 hours before fully maturing. Doctors often measure the “reticulocyte count” in a blood test to determine if the bone marrow is responding correctly to anemia or blood loss.
5. How do vitamins like B12 and Folate (B9) affect blood production?
Both Vitamin B12 and Folate are essential for DNA synthesis. Since erythropoiesis involves rapid cell division, a deficiency in these vitamins leads to “ineffective erythropoiesis.” This results in megaloblastic anemia, where the bone marrow produces abnormally large, fragile cells called macrocytes that die prematurely.
6. What is the difference between Anemia and Polycythemia?
Anemia: A condition where the blood has a low oxygen-carrying capacity. This can be caused by low RBC counts, low hemoglobin levels, or abnormal hemoglobin (as seen in Sickle Cell Disease).
Polycythemia: A condition characterized by an abnormally high number of RBCs. This increases blood viscosity (thickness), which can slow blood flow and increase the risk of blood clots and strokes.
7. Can high altitude affect my red blood cell count?
Yes. At high altitudes, the partial pressure of oxygen is lower, leading to lower oxygen saturation in the blood. The kidneys detect this and increase EPO production, eventually raising your total red blood cell count to compensate. This is why many endurance athletes train at high altitudes.
8. What role does iron play in this process?
Iron is the central component of the heme group within hemoglobin. Each iron atom can bind to one molecule of oxygen. Without sufficient iron, the body cannot produce enough functional hemoglobin, leading to iron-deficiency anemia, characterized by small (microcytic) and pale (hypochromic) red blood cells.
9. What is the “Erythroblastic Island”?
This is a specialized “niche” or microenvironment in the bone marrow where RBCs are born. It consists of a central macrophage (the “nurse cell”) surrounded by developing erythroblasts. The macrophage provides essential nutrients, growth factors, and even “eats” the nuclei expelled by the maturing cells.
10. How are old red blood cells removed from the body?
After about 120 days, the RBC membrane becomes fragile. As these cells try to squeeze through the narrow capillaries of the spleen, they often rupture. Macrophages in the spleen, liver, and bone marrow then break down the remains, recycling the iron for new cells and converting the rest of the heme into bilirubin, which is excreted by the liver.
Erythropoiesis: Definition, Stages, Regulation and Disorders