Intestinal hookworm disease is a significant global health concern, particularly in regions with warm and moist climates. Caused by the nematodes Ancylostoma duodenale and Necator americanus, this infection belongs to the group of Soil-Transmitted Helminths (STH). Hookworm infections primarily affect the small intestine, leading to blood loss, anemia, and protein deficiency, especially in children and adults exposed to contaminated soil. This article explores the biology, life cycle, clinical features, diagnosis, treatment, and prevention strategies of intestinal hookworm infection.
What is Intestinal Hookworm Disease?
Intestinal hookworm disease in humans is caused by two nematode species:
Ancylostoma duodenale
Necator americanus
These parasites are thin, cylindrical worms that attach to the intestinal wall and feed on blood. Infection typically occurs when filariform (L3) larvae, present in contaminated soil, penetrate the skin—most commonly through bare feet. Once inside the body, the larvae migrate to the small intestine, mature into adults, and establish infection.
Hookworm infections are particularly common in areas with poor sanitation and in communities where people walk barefoot.
Habitat and Geographical Distribution
Ancylostoma duodenale and Necator americanus have a global distribution, with the highest prevalence in tropical and subtropical regions where soil remains warm and moist.
Habitat in humans: Small intestine, primarily the distal jejunum
Environmental habitat: Contaminated soil where human feces are present
The parasite thrives in regions with inadequate sanitation, warm temperatures, and high soil moisture, which provide optimal conditions for egg hatching and larval development.
Life Cycle of Intestinal Hookworms
The life cycle of hookworms involves both free-living soil stages and parasitic stages in the human host:
Eggs in Feces: Adult female worms in the small intestine lay eggs, which are passed in the stool.
Larval Development in Soil: Under favorable conditions (moisture, warmth, shade), eggs hatch in 1–2 days into rhabditiform larvae. After 5–10 days and two molts, these larvae develop into infective filariform (L3) larvae. Infective larvae can survive in soil for 3–4 weeks.
Skin Penetration: The L3 larvae penetrate human skin—most often bare feet.
Migration through Body: Larvae enter blood vessels, travel to the heart, and then to the lungs. They penetrate alveoli, ascend the bronchial tree to the pharynx, and are swallowed.
Intestinal Colonization: The larvae reach the jejunum, attach to the intestinal wall, and mature into adult worms. Adults feed on blood, causing anemia and protein loss.
Dormancy (Optional for A. duodenale): Some A. duodenale larvae can become dormant in tissues and reactivate later to establish intestinal infection.
Egg Production: Adult females begin laying eggs, completing the cycle.
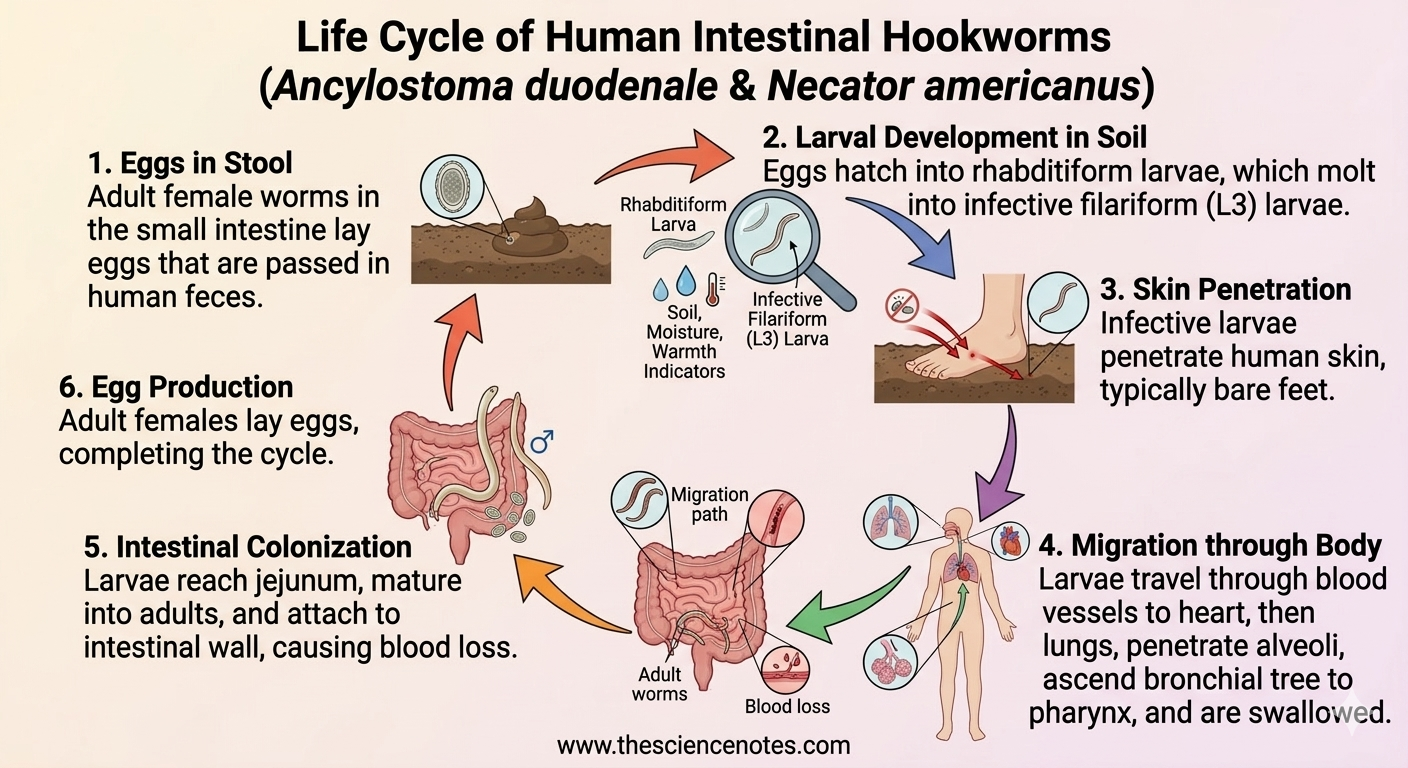
Key point: Adult worms can live for 1–2 years, with some surviving several years under optimal conditions.
Clinical Manifestations
Hookworm infection intensity can vary depending on the number of worms infecting a person. Common symptoms include:
Anemia: Due to chronic blood loss at the site of worm attachment
Protein deficiency: Leading to fatigue, weakness, and stunted growth in children
Gastrointestinal symptoms: Abdominal pain, diarrhea, and loss of appetite
Developmental impact in children: Chronic infection can impair physical growth and cognitive development
High-intensity infections are particularly concerning in school-age children and adults who are repeatedly exposed to contaminated soil.
Laboratory Diagnosis
Diagnosing hookworm infection involves the microscopic identification of eggs in stool samples:
Appearance of eggs: Oval or elliptical, 60 × 40 µm, colorless, thin hyaline shell membrane, not stained with bile.
Techniques: For light infections, a concentration method is often necessary to detect eggs.
Laboratory confirmation helps guide treatment, particularly in areas with high prevalence or mixed infections with other STHs.
Treatment Options
Effective treatment includes anthelmintic medications:
Albendazole (400 mg single dose or 3-day course)
Mebendazole (100 mg twice daily for 3 days)
For patients with anemia, iron supplementation is recommended alongside drug therapy. Regular deworming programs are essential in endemic areas to reduce community burden.
Prevention and Control
Preventing intestinal hookworm infection relies on personal hygiene, protective measures, and environmental sanitation:
Foot Protection: Avoid walking barefoot in areas with potential fecal contamination.
Soil Contact: Minimize direct skin contact with contaminated soil.
Sanitation: Proper disposal of human feces, effective sewage systems, and discouraging open defecation.
Food Safety: Avoid ingestion of soil-contaminated foods.
Mass Deworming Programs: Targeted deworming in schools and high-risk communities can reduce prevalence and prevent reinfection.
By combining personal protection with improved sanitation, communities can significantly reduce the burden of hookworm infection.
Key Takeaways
Intestinal hookworm disease is caused by Ancylostoma duodenale and Necator americanus, transmitted via skin penetration of larvae from contaminated soil.
The parasite resides in the small intestine, feeding on blood and proteins, leading to anemia and nutritional deficiencies.
Diagnosis is primarily through stool microscopy to detect hookworm eggs.
Albendazole and mebendazole are effective treatments, often combined with iron supplementation in anemic patients.
Preventive strategies focus on protective footwear, hygiene, and proper sanitation.
Understanding the life cycle, clinical impact, and preventive measures is crucial for controlling hookworm infection and protecting community health.