Introduction: The New Frontier of Hematologic Oncology
The landscape of cancer treatment has undergone a seismic shift over the last decade. While traditional chemotherapy acts like a broad-spectrum hammer, the emergence of immunotherapy has introduced the precision of a scalpel. Among the most significant breakthroughs in this field is the development of bispecific antibodies. Mosunetuzumab (marketed under the brand name Lunsumio) represents a pinnacle of this innovation.
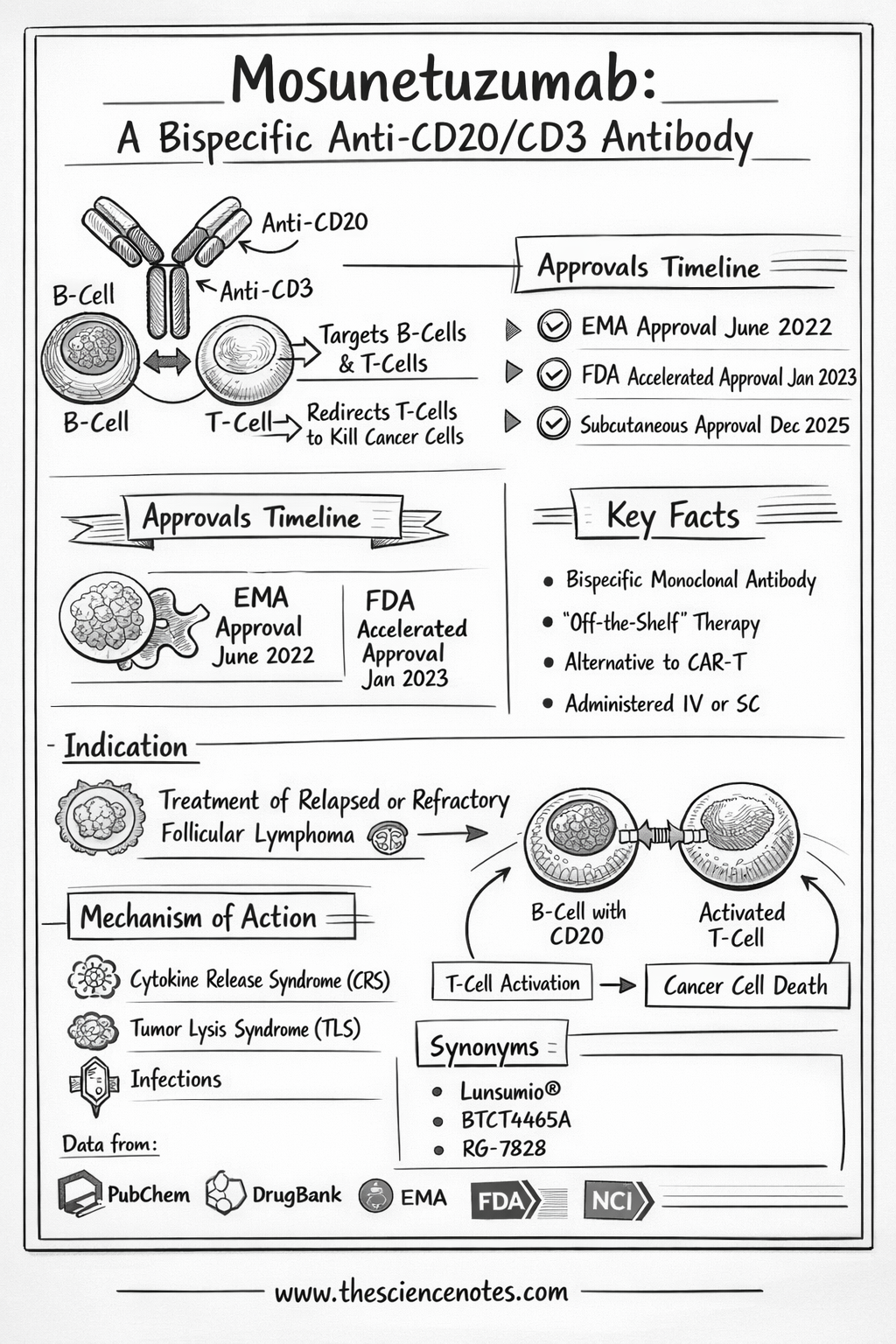
Specifically designed for patients grappling with relapsed or refractory follicular lymphoma (FL), Mosunetuzumab offers a lifeline to those who have exhausted standard lines of treatment. Unlike many other advanced therapies that require complex, patient-specific manufacturing, Mosunetuzumab provides an “off-the-shelf” solution that can be administered quickly, making it a cornerstone of modern hematologic care. This guide provides an exhaustive look at its chemistry, clinical application, and the future of lymphoma management.
What is Mosunetuzumab?
Mosunetuzumab is a humanized IgG1 bispecific monoclonal antibody. To understand its significance, one must first understand the “bispecific” nature of the drug. Traditional monoclonal antibodies, such as Rituximab, target a single antigen. Mosunetuzumab, however, is engineered with two distinct “arms” that allow it to bind to two different targets simultaneously:
CD20: A protein expressed on the surface of B cells, including the malignant cells found in non-Hodgkin lymphomas.
CD3: A protein complex found on T cells, the primary “soldiers” of the human immune system.
By physically linking these two cells, Mosunetuzumab acts as a molecular bridge, forcing the immune system to recognize and destroy cancerous B cells that might otherwise go undetected.
Key Identifiers and Chemical Profile
Generic Name: Mosunetuzumab
Brand Name: Lunsumio
Drug Class: Antineoplastic agent; Bispecific T-cell Engager (BiTE-like)
ATC Code: L01FX25
DrugBank ID: DB15434
Molecular Weight: Approximately 146 kDa
Structure: A sophisticated protein-based therapeutic produced using recombinant DNA technology.
Mechanism of Action: A Deeper Scientific Insight
The brilliance of Mosunetuzumab lies in its conditional activation. In a healthy state, T cells require a complex series of signals to attack a target. Cancer cells often suppress these signals to hide from the immune system. Mosunetuzumab bypasses these evasion tactics through a three-step process:
1. The Immunologic Synapse
When Mosunetuzumab enters the bloodstream, its CD20-binding arm seeks out lymphoma cells, while its CD3-binding arm latches onto nearby T cells. This creates a physical connection known as an immunologic synapse. This proximity is crucial; it brings the T cell close enough to the cancer cell to deliver a lethal blow.
2. Redirection of T-Cell Cytotoxicity
Once the bridge is formed, the T cell undergoes TCR (T-cell receptor) signaling. Crucially, this activation is target-dependent. The T cell does not become “angry” or active until it is successfully bound to the CD20-positive cancer cell. This reduces the risk of systemic, non-specific immune activation.
3. Induced Apoptosis
The activated T cell releases cytotoxic proteins:
Perforins: These molecules punch holes in the membrane of the cancerous B cell.
Granzymes: These enzymes enter through the holes and trigger apoptosis (programmed cell death).
The result is a highly localized “hit” on the tumor, leaving many healthy tissues untouched.
Pharmacodynamics and Clinical Activity
The clinical efficacy of Mosunetuzumab has been rigorously tested in trials such as the GO29781 study. The data suggests that even in patients whose cancer has returned multiple times, Mosunetuzumab can induce deep and lasting remissions.
Impact on the Immune System
Upon administration, clinicians typically observe:
Rapid B-cell Depletion: A significant drop in CD19+ and CD20+ B cells.
Transient Cytokine Elevation: A temporary spike in inflammatory markers (IL-6, IFN-gamma) as the immune system engages.
T-cell Expansion: An increase in the number of activated effector T cells within the tumor microenvironment.
Clinical Response Rates by Subtype
| Lymphoma Type | Overall Response Rate (ORR) | Complete Response (CR) |
| Relapsed/Refractory Follicular Lymphoma | ~80% | ~60% |
| Aggressive NHL (e.g., DLBCL) | ~35-40% | ~20% |
| High-Risk Early Relapse (POD24) | ~75% | ~50% |
These numbers are particularly impressive because many study participants had already failed high-dose chemotherapy or stem cell transplants.
Pharmacokinetics: Absorption, Distribution, and Elimination
Understanding how the body processes Mosunetuzumab is vital for optimizing dosing schedules and managing potential toxicities.
Absorption: When administered via Intravenous (IV) infusion, bioavailability is 100%. Recent shifts toward Subcutaneous (SC) administration show a bioavailability of approximately 75–80%, offering a more convenient delivery method for patients.
Distribution: With a volume of distribution (Vd) of roughly 5.49 L, the drug primarily circulates in the vascular and interstitial spaces, ensuring it reaches lymph nodes where tumors reside.
Metabolism: Like most monoclonal antibodies, Mosunetuzumab is not processed by the cytochrome P450 enzymes in the liver. Instead, it is broken down into small peptides and amino acids through general protein catabolism.
Elimination and Half-Life: The terminal half-life is approximately 16 days. This long duration allows for a convenient dosing schedule, typically once every 21 days (a 3-week cycle).
Clinical Use and Indications
As of early 2026, the primary indication for Mosunetuzumab is for adult patients with relapsed or refractory follicular lymphoma (FL) who have received at least two prior systemic therapies.
Why Follicular Lymphoma?
FL is often considered “indolent” but is characterized by a pattern of repeated relapses. With each subsequent relapse, the duration of remission usually shortens. Mosunetuzumab provides a potent option for these “late-line” patients, often achieving a Complete Response (CR) where the cancer becomes undetectable.
Emerging Research
Research is currently expanding into:
First-line treatment in combination with CHOP chemotherapy.
Chronic Lymphocytic Leukemia (CLL).
Diffuse Large B-Cell Lymphoma (DLBCL) as a bridge to other therapies.
Why Mosunetuzumab is a Breakthrough Therapy
1. Overcoming “Rituximab Resistance”
Many B-cell cancers eventually stop responding to Rituximab because the cancer cells find ways to hide their CD20 proteins or avoid antibody-dependent cellular cytotoxicity (ADCC). Mosunetuzumab is more potent because it doesn’t just “flag” the cell for the immune system; it manually drags a T cell to the site, bypassing many resistance mechanisms.
2. The “Off-the-Shelf” Advantage over CAR-T
While CAR-T cell therapies (like Yescarta or Kymriah) are revolutionary, they require:
Leukapheresis (extracting a patient’s blood).
Weeks of lab manufacturing.
High costs and intensive hospitalization.
Mosunetuzumab is ready-to-use. A patient can be diagnosed with a relapse on Monday and begin Mosunetuzumab treatment on Tuesday.
Administration and “Step-Up” Dosing
To ensure patient safety, Mosunetuzumab is administered using a step-up dosing schedule during the first cycle. This “primes” the immune system and prevents an overreaction.
Cycle 1, Day 1: 1 mg (Initial small dose)
Cycle 1, Day 8: 2 mg (Intermediate dose)
Cycle 1, Day 15: 60 mg (Full therapeutic dose)
Subsequent Cycles: 30 mg or 60 mg every 21 days depending on the protocol.
This gradual increase is specifically designed to mitigate the risk of Cytokine Release Syndrome (CRS).
Safety and Adverse Effects
While generally better tolerated than intensive chemotherapy, Mosunetuzumab does carry specific risks associated with T-cell activation.
Cytokine Release Syndrome (CRS)
CRS occurs when activated T cells release a flood of inflammatory cytokines.
Symptoms: Fever (most common), chills, low blood pressure (hypotension), and hypoxia.
Management: Most cases are Grade 1 or 2 and are managed with fluids, oxygen, or the drug Tocilizumab.
Neurotoxicity (ICANS)
Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) is rarer with Mosunetuzumab than with CAR-T but can still occur.
Signs: Confusion, altered consciousness, or “dysgraphia” (difficulty writing).
Monitoring: Patients are often asked to perform simple tasks, like writing a sentence daily, to check for early neurological changes.
Other Side Effects
Neutropenia: A drop in white blood cell counts, increasing infection risk.
Hypogammaglobulinemia: Lower levels of antibodies, which may require IVIG (Intravenous Immunoglobulin) replacement.
Tumor Lysis Syndrome (TLS): Rapid killing of cancer cells can release toxins into the blood, potentially harming the kidneys.
Drug Interactions and Special Populations
Interactions
Mosunetuzumab does not have traditional “drug-drug” interactions in the way small molecules do. However, the systemic inflammation caused by the drug can transiently suppress CYP450 enzymes. Caution is advised with drugs with narrow therapeutic indices, such as Warfarin (Coumarin) or Phenytoin.
Vaccinations
Patients should avoid live-attenuated vaccines (e.g., Yellow Fever, MMR) during treatment, as the suppressed B-cell environment could lead to vaccine-derived infections.
Special Populations
Pregnancy: Based on its mechanism, Mosunetuzumab can cause fetal B-cell depletion. Effective contraception is required during treatment and for 3 months after the final dose.
Geriatric Use: Clinical trials included a significant number of patients over 65, showing no major differences in safety or efficacy compared to younger patients.
Future Perspectives: Beyond the Third Line
The future of Mosunetuzumab is bright. We are moving toward fixed-duration therapy, where patients receive treatment for a set number of cycles (e.g., 8 to 17 cycles) and then stop, rather than staying on the drug indefinitely. This “treatment holiday” improves quality of life and reduces long-term toxicity.
Furthermore, combining Mosunetuzumab with other agents like Polatuzumab Vedotin or Lenalidomide is showing promise in creating even more durable remissions for the most aggressive forms of lymphoma.
Comparative Overview: Mosunetuzumab vs. CAR-T vs. Rituximab
The following table compares these therapies specifically in the context of Follicular Lymphoma (FL) as of early 2026.
Comparative Overview: Mosunetuzumab vs. CAR-T vs. Rituximab
| Feature | Rituximab (Rituxan) | Mosunetuzumab (Lunsumio) | CAR-T (e.g., Yescarta / Breyanzi) |
| Drug Class | Monoclonal Antibody (Anti-CD20) | Bispecific T-cell Engager (CD20 x CD3) | Gene-engineered T-cell Therapy |
| Mechanism | Flags B-cells for the native immune system to find. | Physically bridges T-cells to B-cells to force a kill. | Reprograms patient’s own T-cells to hunt cancer. |
| Target Line | 1st line and maintenance. | 3rd line+ (Relapsed/Refractory). | 3rd line+ (Relapsed/Refractory). |
| Availability | Immediate (“Off-the-shelf”). | Immediate (“Off-the-shelf”). | Custom-made (requires 3–5 week wait). |
| Administration | IV or Subcutaneous (Quick). | IV or Subcutaneous (Step-up dosing). | One-time IV infusion (after chemo). |
| Hospitalization | Not required (Outpatient). | Generally Outpatient (Cycle 1). | Often required (Inpatient monitoring). |
| Efficacy (ORR) | High in early lines (~70-80%). | Very High in R/R (~80%). | Extremely High (~90%+). |
| Durability | Varies; resistance common over time. | High; deep remissions (CR ~60%). | Potential for long-term “cure.” |
| CRS Risk | Very Low (Infusion reactions only). | Moderate (Common but mostly low-grade). | High (Can be severe/Grade 3+). |
| Est. Drug Cost | ~$30k – $50k per year. | ~$180k (Fixed-duration course). | ~$400k – $450k (One-time). |
Conclusion
Mosunetuzumab (Lunsumio) represents a paradigm shift in cancer treatment. By combining the precision of monoclonal antibodies with the raw power of T cells, it offers a “best-of-both-worlds” approach to treating follicular lymphoma. Its “off-the-shelf” availability and manageable safety profile make it an accessible, life-extending option for patients who once had few places to turn.
As our understanding of the immune microenvironment grows, Mosunetuzumab will likely serve as a blueprint for future bispecific therapies across various types of cancer.
Disclaimer: This content is for educational purposes only and should not replace professional medical advice. Always consult your healthcare provider for treatment decisions.