Proteins are the most versatile and functionally diverse macromolecules in the biological world. While DNA holds the blueprint for life, proteins are the actual laborers that execute the instructions. However, a protein is not just a string of chemical components; it is a sophisticated molecular machine whose power is derived entirely from its shape.
The process by which a linear chain of amino acids transforms into a complex, three-dimensional masterpiece is known as protein folding. Understanding this process is fundamental to modern biochemistry, as the “form follows function” rule dictates every breath we take, every beat of our heart, and even how our bodies fight off infection.
1. The Fundamental Unit: Amino Acids and Polypeptides
At the most basic level, proteins are polymers constructed from 20 different monomers called amino acids. Each amino acid shares a common core structure: a central carbon atom ($\alpha$-carbon) bonded to a hydrogen atom, an amino group ($-NH_2$), a carboxyl group ($-COOH$), and a unique side chain known as the R-group.
The Synthesis of Polypeptides
During the process of translation in the ribosome, amino acids are joined together via peptide bonds. This covalent bond forms through a dehydration synthesis reaction between the carboxyl terminus of one amino acid and the amino terminus of the next.
The resulting chain is called a polypeptide. While the terms “polypeptide” and “protein” are often used interchangeably in casual conversation, scientists distinguish them by their state: a polypeptide is the raw chemical chain, whereas a protein is a polypeptide that has folded into its functional, biologically active 3D conformation.
2. The Hierarchical Levels of Protein Structure
To manage the immense complexity of these molecules, scientists describe protein structure through four distinct hierarchical levels.
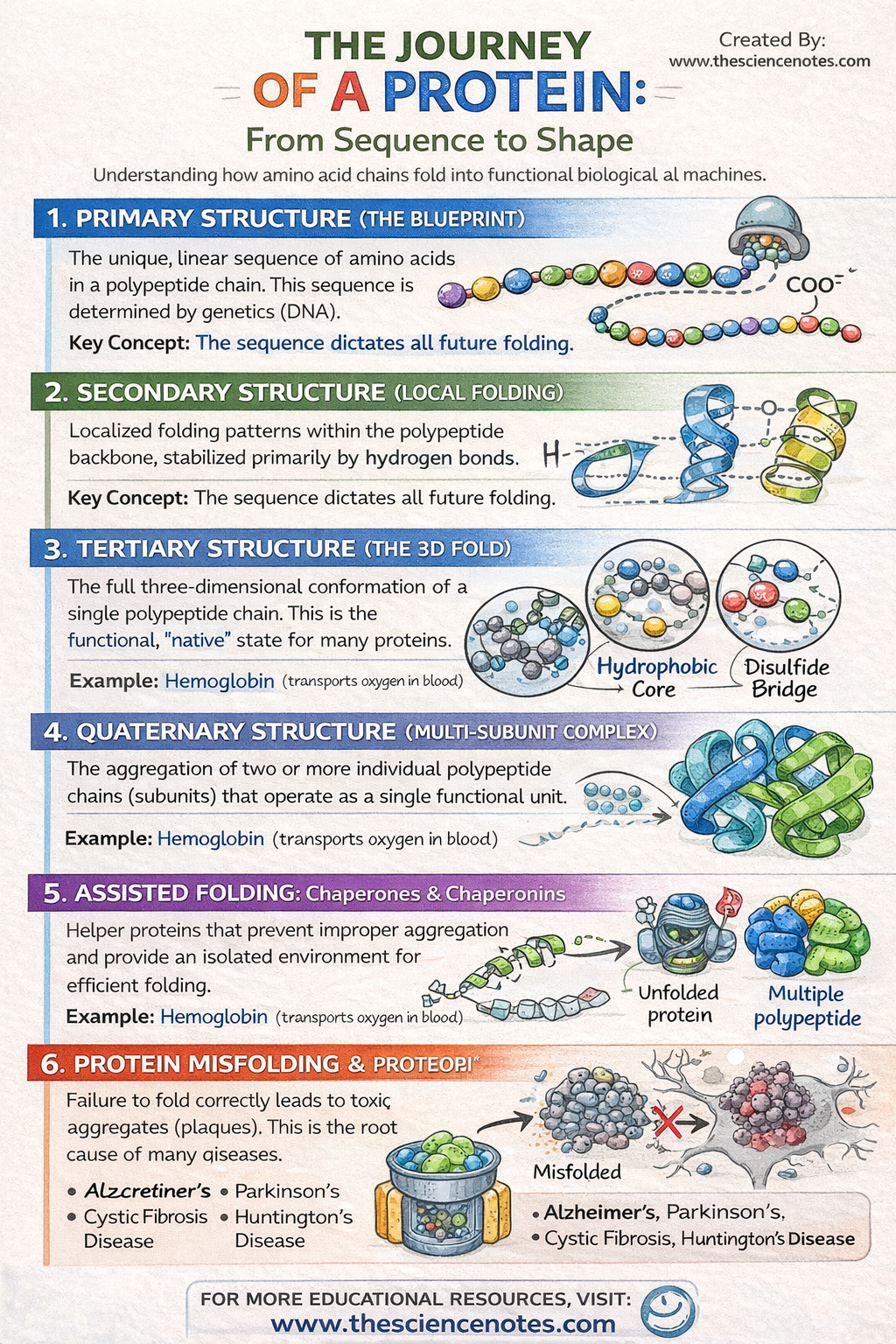
I. Primary Structure: The Genetic Blueprint
The primary structure is simply the linear sequence of amino acids. Despite its simplicity, this sequence is the most critical determinant of the protein’s future. The specific order of amino acids is dictated by the DNA sequence of the corresponding gene. Because each of the 20 amino acids has different chemical properties (size, charge, and hydrophobicity), their arrangement determines exactly how the chain will eventually attract or repel itself to form a 3D shape.
II. Secondary Structure: Localized Folding
As the polypeptide emerges from the ribosome, it begins to form localized “neighborhoods” of shape. These are stabilized by hydrogen bonds between the atoms of the polypeptide backbone (not the side chains).
Alpha-Helices ($\alpha$-helices): A delicate, coil-like spiral held together by hydrogen bonds between every fourth amino acid.
Beta-Pleated Sheets ($\beta$-sheets): Two or more segments of the chain lying side-by-side, connected by hydrogen bonds to form a rigid, sheet-like structure.
III. Tertiary Structure: The Global 3D Fold
This level represents the final “native conformation” for most single-chain proteins. While the secondary structure is about the backbone, the tertiary structure is all about the R-group interactions. This is where the protein collapses into a globular or fibrous shape based on the chemistry of its side chains.
IV. Quaternary Structure: Multi-Unit Assemblies
Some of the most complex proteins, such as hemoglobin or DNA polymerase, consist of multiple polypeptide chains (subunits) that must come together to function. This assembly is the quaternary structure. Without the correct arrangement of these subunits, the protein remains inactive.
3. The Forces That Drive Folding
Protein folding is a “search” for the most thermodynamically stable state. Several key chemical forces act as the “engineers” of this process:
The Hydrophobic Effect
This is perhaps the most significant force in protein folding. In the watery environment of the cell, non-polar (hydrophobic) amino acid side chains naturally want to avoid water. As the protein folds, these hydrophobic residues cluster together in the interior “core” of the protein, while polar and charged (hydrophilic) residues remain on the exterior to interact with water.
Molecular “Staples”: Disulfide Bonds
Cysteine is a unique amino acid because its side chain contains a sulfur-containing thiol group ($-SH$). When two cysteines are brought close together during folding, they can form a covalent disulfide bridge. These act like molecular staples, locking the protein into its final, most stable shape and protecting it from being easily unfolded.
Van der Waals and Electrostatic Forces
Van der Waals Forces: Once the hydrophobic core is tightly packed, these weak attractions between atoms provide an extra layer of structural stability.
Ionic Bonds (Salt Bridges): Positively charged side chains (like Lysine) can attract negatively charged ones (like Aspartic Acid) to “zip” parts of the protein together.
4. Molecular Chaperones: The Quality Control Team
For a long time, scientists believed proteins folded entirely on their own (Anfinsen’s Dogma). However, we now know that the cellular environment is too crowded for most proteins to fold successfully without help. Enter molecular chaperones.
Chaperonins: These are barrel-shaped protein complexes that act as “safe rooms.” An unfolded polypeptide enters the barrel, a “lid” closes, and the protein is allowed to fold in isolation, away from other molecules that might cause it to clump or aggregate.
Heat Shock Proteins (HSPs): These proteins increase in concentration when the cell is stressed by heat. They bind to exposed hydrophobic regions of unfolding proteins to prevent them from sticking to each other and forming toxic “clumps.”
The Role of Molecular Chaperones: The Quality Control Team
For a long time, it was believed that proteins folded entirely on their own based solely on their sequence (Anfinsen’s Dogma). However, the interior of a cell is a crowded, “salty soup” of organelles and other macromolecules. In this environment, newly synthesized polypeptides are at high risk of clumping together (aggregating) or folding into “dead-end” shapes that offer no biological utility.
To ensure survival, cells have evolved a sophisticated quality control system led by molecular chaperones. These proteins do not dictate the final shape of the protein—the amino acid sequence still does that—but they provide the assistance and environment necessary for the protein to find its “native conformation” efficiently.
1. Chaperonins: The Isolation Chambers
Chaperonins, such as the well-studied GroEL/GroES complex in bacteria, are barrel-shaped protein structures. They act as “safe rooms” for folding.
Mechanism: An unfolded or partially folded polypeptide enters the central cavity of the “barrel.”
Isolation: A “lid” (chaperonin cap) closes the chamber. Inside this protected microenvironment, the protein is shielded from the crowded cytoplasm.
Folding: The environment inside the barrel often has chemical properties that favor correct folding. Once the process is complete, the lid opens, and the functional protein is released.
2. Heat Shock Proteins (HSPs): The Molecular Bodyguards
Heat shock proteins, such as Hsp70, are the cell’s first line of defense against misfolding, especially during environmental stress like high fever or pH changes.
Mechanism: They identify and bind to exposed hydrophobic regions on an unfolded polypeptide.
Prevention: By “masking” these sticky hydrophobic patches, HSPs prevent the polypeptide from sticking to other proteins in the cell.
Release: Using energy from ATP, the HSP eventually releases the protein, giving it another chance to fold correctly.
Comparison: Chaperonins vs. Heat Shock Proteins
While both are chaperones, they operate at different stages of the protein’s life cycle.
| Feature | Heat Shock Proteins (e.g., Hsp70) | Chaperonins (e.g., GroEL/ES) |
| Physical Shape | Small, clamp-like proteins. | Large, barrel-shaped complexes. |
| Primary Action | Binds to and stabilizes “sticky” regions. | Provides an isolated “cage” for folding. |
| Timing | Often acts early, while the protein is being made. | Acts later, on partially folded intermediates. |
| Energy Use | Requires ATP to bind/release the protein. | Requires ATP to close the lid and cycle the barrel. |
| Goal | Prevents aggregation and “misfolding” during stress. | Facilitates the final 3D “native” fold. |
Enzymatic Helpers: PDI and PPI
In addition to chaperones, specific enzymes speed up the chemical “locking” of a protein:
Protein Disulfide Isomerase (PDI): This enzyme is critical for proteins that require disulfide bonds. It helps the protein rapidly “test” different bond combinations until the most stable, correct disulfide bridges are formed.
Peptidyl Prolyl Isomerase (PPI): This enzyme helps rotate bonds involving the amino acid Proline, which is often a “kink” in the chain that can slow down the folding process.
Without this team of chaperones and enzymes, the “folding funnel”—the path a protein takes to find its stable shape—would be too slow and prone to errors, leading to the cellular “trash” that causes neurodegenerative diseases.
5. Architectural Diversity: Globular vs. Fibrous
Proteins generally fall into two broad structural categories based on their tertiary or quaternary shapes:
Globular Proteins
These are spherical, compact, and generally soluble in water. Their surfaces are covered in hydrophilic residues, making them perfect for moving through the bloodstream or cytoplasm.
Examples: Hemoglobin (oxygen transport), Insulin (hormone signaling), and almost all enzymes (catalysis).
Fibrous Proteins
These are long, rope-like, and insoluble in water. They are built for strength and durability rather than chemical reactivity.
Examples: Keratin (strengthening hair and skin), Collagen (providing structure to tendons and bone), and Actin/Myosin (facilitating muscle movement).
6. When Folding Goes Wrong: Denaturation and Disease
Since a protein’s function is purely dependent on its shape, losing that shape—a process called denaturation—is usually catastrophic.
Causes of Denaturation
Heat: Increases kinetic energy, vibrating the protein until weak hydrogen bonds break.
pH Changes: Disrupts the ionic bonds (salt bridges) by changing the charge of the side chains.
Chemicals: Urea or detergents can disrupt the hydrophobic core.
Proteopathy: The Diseases of Misfolding
If a protein misfolds and the cell’s quality control systems (like chaperones) fail to fix or destroy it, these proteins can aggregate into “amyloid plaques.” These plaques act like “molecular sand” in the gears of the cell, eventually leading to cell death. This is the underlying mechanism for many neurodegenerative conditions:
Alzheimer’s Disease: Caused by the accumulation of beta-amyloid plaques.
Parkinson’s Disease: Linked to the misfolding of alpha-synuclein.
Cystic Fibrosis: Caused by a single amino acid deletion that prevents a membrane protein from folding correctly, leading to its destruction by the cell before it can ever function.
Conclusion: The Precision of Biological Engineering
The journey of a protein from a simple genetic sequence to a functional 3D machine is one of the most remarkable feats of biological engineering. Every interaction—from the strength of a covalent disulfide bond to the subtle “shyness” of a hydrophobic residue—is perfectly balanced to ensure the protein can perform its life-sustaining role. As we continue to map the “proteome,” our understanding of these folding pathways will unlock new treatments for diseases and allow us to design synthetic proteins that could solve global challenges in medicine and industry.