Ion channels are integral membrane proteins that regulate the movement of ions across cellular membranes, thereby generating electrical signals and controlling numerous physiological processes. They play a fundamental role in neuronal excitability, muscle contraction, and intracellular signaling pathways. Ion channels function both as signal generators and effectors, particularly through calcium-mediated signaling networks. This review provides a comprehensive yet concise overview of ion channel structure, classification, and functional mechanisms, with a focus on voltage-gated and ligand-gated ion channels and their roles in cellular communication and signaling.
Introduction
Ion channels are specialized transmembrane proteins that span the lipid bilayer of cell membranes and facilitate the selective passage of ions such as sodium (Na⁺), potassium (K⁺), calcium (Ca²⁺), and chloride (Cl⁻). These proteins are essential for establishing and maintaining electrochemical gradients, which are critical for cellular homeostasis and signaling.
Ion channel signaling originates at the plasma membrane in response to various stimuli, including changes in membrane potential, ligand binding, and mechanical stress. Structurally, ion channels are hetero-oligomeric complexes composed of noncovalently bonded subunits that form aqueous pores. Conformational changes within these protein structures regulate channel opening and closing, thereby controlling ion flux.
Functionally, ion channels serve dual roles. First, they act as signal generators, particularly through Ca²⁺ signaling pathways, where controlled ion entry and release generate intracellular signals. Second, they function as effectors, responding to intracellular messengers and participating in downstream signaling cascades. These properties make ion channels central to processes such as neurotransmission, hormone secretion, and immune cell activation.
Classification of Ion Channels
Ion channels are broadly categorized into two main types based on their mode of activation:
1. Voltage-Gated Ion Channels (VGICs)
Voltage-gated ion channels are activated by changes in membrane potential and are critical for electrical signaling in excitable cells such as neurons and muscle fibers. These channels open when the membrane potential reaches a specific threshold, allowing ions to move along their electrochemical gradients.
VGICs typically exist in three functional states:
- Closed state – channel is not conducting ions
- Open state – ions flow through the channel
- Inactivated state – channel is temporarily non-conductive despite stimulus
Based on ion selectivity, VGICs are classified into sodium (Na⁺), potassium (K⁺), calcium (Ca²⁺), and chloride (Cl⁻) channels.
2. Ligand-Gated Ion Channels (LGICs)
Ligand-gated ion channels are activated by the binding of specific ligands, such as neurotransmitters or hormones. These channels play a central role in synaptic transmission by converting chemical signals into electrical responses.
Ligand binding induces conformational changes that open the channel pore, allowing selective ion passage. This results in changes in membrane potential, leading to processes such as action potential generation, muscle contraction, and hormone release.
Structure and Function of Voltage-Gated Ion Channels
The Voltage-gated ion channels are complex transmembrane proteins with highly specialized structures that enable voltage sensing and ion selectivity.
Voltage-Gated Sodium Channels
The Voltage-gated sodium channels consist of a single polypeptide chain organized into four homologous domains (I–IV). Each domain contains six transmembrane α-helices. The fourth segment (S4) in each domain contains positively charged residues, such as arginine, which act as voltage sensors.
These channels are essential for:
- Initiation of action potentials
- Rapid depolarization of excitable cells
- Signal propagation along neurons and muscle fibers
Accessory β-subunits associate with the α-subunit and modulate channel kinetics and localization.
Voltage-Gated Potassium and Calcium Channels
Voltage-gated potassium channels are typically tetrameric, consisting of four identical or similar α-subunits. They play a key role in repolarizing the membrane following action potentials.
Voltage-gated calcium channels share structural similarity with sodium channels and include additional auxiliary subunits such as α2δ, β, and γ. These channels regulate calcium entry into cells, which is critical for neurotransmitter release, gene expression, and intracellular signaling.
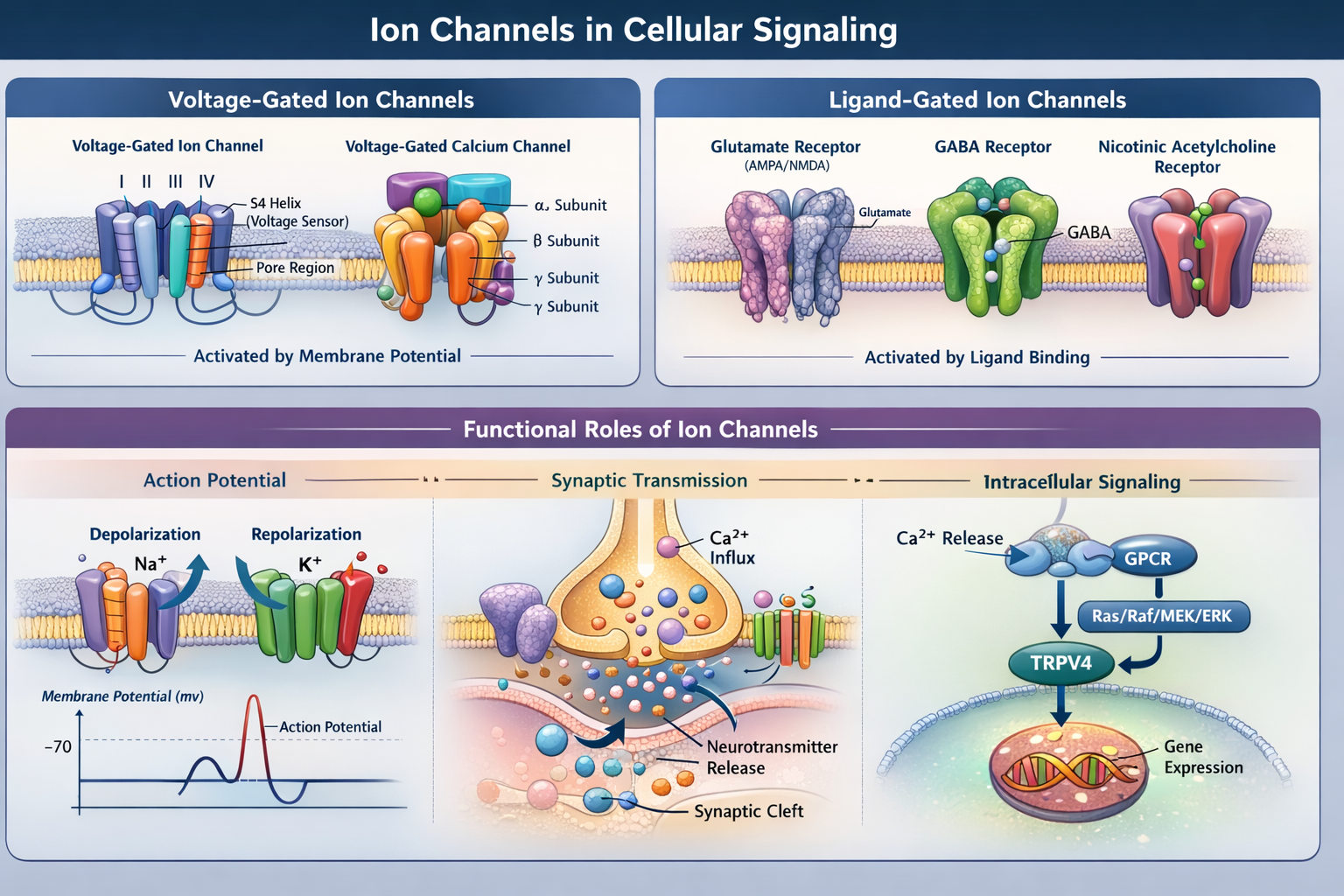
Role of Ion Channels in Action Potential Generation
Ion channels are fundamental to the generation and propagation of action potentials, the electrical signals used by neurons to communicate.
At rest, the neuronal membrane potential is approximately −70 mV. Upon stimulation:
- Depolarization: Voltage-gated Na⁺ channels open, allowing rapid Na⁺ influx.
- Action potential generation: This influx causes a rapid rise in membrane potential.
- Calcium influx: At the presynaptic terminal, Ca²⁺ entry triggers synaptic vesicle fusion and neurotransmitter release.
- Repolarization: Na⁺ channels inactivate, and K⁺ channels open, allowing K⁺ efflux and restoring resting potential.
This coordinated sequence of events ensures efficient signal transmission across neurons.
Ligand-Gated Ion Channels and Synaptic Transmission
Ligand-gated ion channels mediate fast synaptic communication by responding to neurotransmitter binding.
Glutamate Receptors
Glutamate is the primary excitatory neurotransmitter in the central nervous system. Ionotropic glutamate receptors include AMPA and NMDA receptors.
- AMPA receptors: Mediate rapid Na⁺ influx and fast excitatory postsynaptic potentials
- NMDA receptors: Permit Ca²⁺ entry and are involved in synaptic plasticity and memory formation
NMDA receptor activation requires both ligand binding and removal of a Mg²⁺ block, making it a coincidence detector for synaptic activity.
Inhibitory and Cholinergic Receptors
Other ligand-gated channels include:
- GABA receptors: Mediate inhibitory neurotransmission via Cl⁻ influx
- Glycine receptors: Also inhibitory, primarily in the spinal cord
- Nicotinic acetylcholine receptors (nAChRs): Mediate excitatory signaling at neuromuscular junctions
Structure and Mechanism of Nicotinic Acetylcholine Receptors
The nicotinic acetylcholine receptor is a well-studied ligand-gated ion channel that illustrates the principles of channel gating.
Structural Organization
- Pentameric structure composed of α, β, γ, and δ subunits
- Ligand-binding sites located at subunit interfaces
Activation Mechanism
In the absence of acetylcholine, the channel remains closed due to hydrophobic residues lining the pore. Upon ligand binding:
- Conformational changes occur
- Hydrophobic residues are replaced with polar residues
- The pore widens, allowing ion passage
This transition from closed to open state enables Na⁺ and Ca²⁺ influx, leading to membrane depolarization and muscle contraction.
Ion Channels in Intracellular Signaling Pathways
Beyond their role in electrical signaling, ion channels are integral components of intracellular signaling networks.
Calcium channels, in particular, are central to signal transduction. Changes in intracellular Ca²⁺ concentration regulate processes such as gene expression, enzyme activation, and cell proliferation.
Recent studies have demonstrated interactions between ion channels and major signaling pathways:
- Epidermal growth factor (EGF) signaling activates calcium channels via the Ras/Raf/MEK/ERK pathway
- TRPV4 channels interact with G protein-coupled receptors (GPCRs), contributing to vascular function and inflammation
These findings highlight the integration of ion channel activity with broader cellular signaling mechanisms.
Physiological and Pathological Significance
Ion channels are essential for normal physiological function, and their dysregulation is associated with numerous diseases, collectively termed channelopathies.
Examples include:
- Neurological disorders (epilepsy, migraine)
- Cardiac arrhythmias
- Muscle disorders
- Pain syndromes
Understanding ion channel structure and function has enabled the development of targeted pharmacological agents, including channel blockers and modulators used in treating cardiovascular and neurological diseases.
Conclusion
Ion channels are fundamental to cellular physiology, mediating ion transport, electrical signaling, and intracellular communication. Their diverse structures and activation mechanisms enable precise control of critical biological processes. Advances in structural biology, electrophysiology, and pharmacology continue to enhance our understanding of ion channel function and their role in disease, providing new opportunities for therapeutic intervention.