Author : Binod GC
T cells are lymphocytes that play a variety of activities in the immune system, including pathogen eradication and tumor surveillance. They play a crucial role in the adaptive immune response. The -chain and -chain of the TCR form a heterodimer that allows T cells to bind to specific ligands. The clonal identity of each T cell is provided by the rearrangement of genomic DNA throughout development, which provides each T cell with a TCR of unique specificity. TCRs play a role in T cell activity and the formation of the immunological synapse by connecting T cells to antigen-presenting cells (APC). T cells are constantly scanning lymphoid and peripheral tissues for antigens such as peptides or lipids displayed by other cells’ major histocompatibility complex (pMHC) molecules(Sewell 2012). TCR activation is triggered by MHC molecules on antigen presenting cells (APC), which present antigen peptides to TCR complexes and initiate a series of cellular signaling cascades that result in cellular proliferation, differentiation, cytokine production, and/or activation-induced cell death( Gorentla and Zhong 2012). T cell signaling pathway.
Lck, a Src-family protein tyrosine kinase, is first activated, resulting in phosphorylation of CD3 coreceptor cytoplasmic domains, specifically at immunoreceptor tyrosine-based activation motifs (ITAMs). These phosphorylated ITAMs on CD3-zeta act as binding sites for ZAP-70, a zeta chain-associated protein kinase(Guirado, de Aós et al. 2002, Gorentla and Zhong 2012). Lck-mediated phosphorylation and subsequent autophosphorylation activates ZAP-70. Activated ZAP-70 then phosphorylates a number of linker/adapter proteins, including the linker for T-cell activation (LAT) and the Src-homology 2 domain-containing 76-kDa leukocyte protein (SLP-76)(Koretzky and Boerth 1999, Gorentla and Zhong 2012). Following that, additional signaling proteins are recruited, resulting in calcium mobilization, actin cytoskeleton reorganization, and Ras guanosine triphosphate hydrolase activation (GTPases).
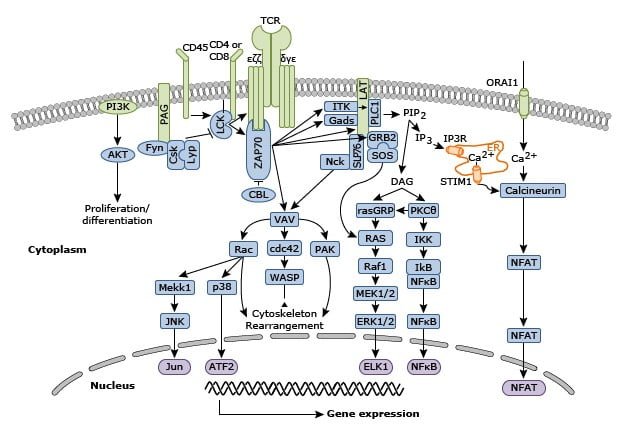
Downstream from these events, transcription factors such as nuclear factor of activated T cells (NF-AT), activator protein 1 (AP-1) and nuclear factor kappa light-chain enhancer of activated B cells (NF-kappa-B) are activated, causing changes in the pattern of T cell gene expression, proliferation, and effector function differentiation.
TCR/CD3 Complex
TCR is made up of six different protein molecules that combine to form the TCR heterodimer that is capable of recognizing ligands. When CD3 molecules (CD3, CD3, and CD3) bind to the TCR heterodimer, they form a unique sequence motif for tyrosine phosphorylation known as immuno-receptor tyrosine-based activation motifs (ITAMs). TCR polypeptides have extremely short cytoplasmic tails, and all proximal signaling events are mediated by CD3 molecules. The interaction of the TCR/CD3 complex is critical in facilitating cell sufficiency such as receptor assembly, cell-surface expression, and signaling(van Boxel, Holmes et al. 2010).
TCR Activation
A number of co-stimulatory receptors regulate TCR activation. Many transmembrane receptors have an impact on various aspects of TCR signaling. CD28 promotes TCR-induced proliferation and differentiation of naive T cells. When CD28 binds to B7-1 or B7-2, it provides an initial adhesion ability to the T-cell by bringing the T-cell and APC membranes together. Furthermore, CD45 regulates TCR signaling by modulating the phosphorylation state of tyrosine kinases like Lck and Fyn, as well as antagonizing the inhibitory effect of inhibitory proteins, promoting T-cell activation.
Distal TCR signaling
TCR activation results in proximal signaling, which is followed by activation of multiple distal pathways, including Ca2+–calcineurin–NFAT, PKC–IKK–NF, RASGRP1–RAS–ERK1/2, and TSC1/2–mTOR, with the assistance of secondary messengers, enzymes, and numerous adaptor proteins. Phospholipase C1 ((PLCγ1) is the primary molecule involved in the downstream signaling cascade after TCR activation. Phosphatidylinositol 4,5-bisphosphate (PIP2) in the membrane is hydrolyzed into diacylglycerol (DAG) and inositol-3-phosphate by activated PLCγ1 (IP3). These two messengers start the distal signaling cascade, which is necessary for T cell activation. PKCθ, RASGRP1, and PDK1-mediated pathways are activated, whereas IP3 activates a Ca2+-dependent calcineurin NFAT pathway.
Ca2+calcineurin NFAT pathway
Phospholipase Cγ1 (PLCγ1) is the main molecule contributing to the downstream signaling cascade following TCR activation. Phosphatidylinositol 4,5-bisphosphate (PIP2) present in the membrane is hydrolyzed by activated PLCγ1 into diacylglycerol (DAG) and inositol-3-phosphate (IP3). These two messengers initiate distal signaling cascade which is important for T cell activation. PKCθ, RASGRP1, and PDK1-mediated pathways whereas IP3 prompts the initiation of a Ca2+-dependent calcineurin NFAT pathway.
PIP2 produces IP3, which adheres to Ca2+-permeable ion channel receptors (IP3R) on the endoplasmic reticulum (ER), enabling Ca2+ to access the cytoplasm. The expression of stromal interaction molecule (STIM), a transmembrane protein, can sense intracellular Ca2+ levels in the ER. When the level falls, the Ca2+ influx into T cells is provoked by the Orai1 type plasma membrane calcium-release enabled calcium (CRAC) channel. When intracellular Ca2+ rises, it activates calcineurin, a protein phosphatase which dephosphorylates the nuclear factor of activated T cells (NFAT), causing it to translocate to the nucleus. In the nucleus, NFAT interacts with AP-1 transcriptional factors (JUN/FOS) derived from the DAG–RAS–MAPK–ERK1/2 pathway(Beach, Gonen et al. 2007).
This complex stimulates the expression of several genes, including IL2 and other molecules required for T cell activation. When AP1 is absent, NFAt alone activates genes responsible for ubiquitin ligases and diacylglycerol kinase (DGK), which are responsible for T cell anergy, a state of T cell unresponsiveness, which is one of the processes that induce immune tolerance. Thus, NFAT proteins regulate two opposing T cell functions: activation and anergy(Macian 2005).
Ca2+ activates a Ca2+/calmodulin-dependent kinase (CaMK) that mediates T cell activation by activating transcription factors like cyclic-AMP-responsive-element-binding protein and myocyte enhancer factor 2. In humans, a missense mutation in Orai1 causes impaired Ca2+ signaling, which affects nuclear translocation of NFAT and thus NFAT-induced cytokines production, resulting in severe combined immunodeficiency (SCID)(Krappmann, Emmerich et al. 1999). As a result, the universal secondary messenger Ca2+ regulates several important functions in T cells, such as proliferation, differentiation, and cytokine production.
PKCθ–IKK–NF-κβ pathway
PKCθ is a novel isoform of the Protein Kinase C (PKC) family that binds to DAG via the PKCθ conserved region 1 (C1) domain, which is required for recruitment to the lipid raft after TCR engagement. Despite the fact that T cells express several other PKCs, PKCθ plays important roles in T cell activation. Activation of PKCθ after TCR activation corresponds to the formation of a complex of three molecules of adaptor protein in the cytoplasm called CBM complex consisting of caspase recruitment domain-containing membrane-associated guanylate kinase protein-1 (CARMA1), B cell lymphoma/leukemia 10 (BCL10), and mucosa-associated lymphoid tissue translocation protein-1 (MALT1). PKCθ activation phosphorylates CARMA1, which is required for oligomerization and association with BCL10. When MALT1 binds to BCL10, it recruits an E3 ubiquitin ligase known as tumor necrosis factor receptor-associated factor 6 (TRAF6), which polyubiquitinates and degrades IKK, or the NF- essential modifier (NEMO), a regulatory protein of the IKK complex. The catalytic subunits of I kinases (IKK), and, are no longer inhibited, and they phosphorylate I, causing it to be ubiquitinated and degraded. As a result, NF- is released from its inhibitory I complex in the cytoplasm and translocate into the nucleus to regulate gene expression(Krappmann, Emmerich et al. 1999).
RASGRP1–RAS–ERK1/2–AP1 pathway
DAG activates and recruits a RAS guanyl nucleotide-releasing protein (RASGRP1) to the plasma membrane. Son of Sevenless (Sos) and RASGRP1 are two well-known guanine exchange factors (GEFs) that are responsible for RAS activation in T cells. RAS binds to GTP and activates the serine/threonine kinase Raf1 to initiate the RAS-MAPK cascade. Raf1 is a MAPK that activates MAPKK, which then activates MAPKKK like MEK1/2, which phosphorylates and activates MAPK extracellular signal-regulated kinase-1 and 2 (ERK1/2). ERK1/2 signaling regulates T cell development, differentiation, and TCR-induced signal strength. ERKs also cause the phosphorylation and activation of their downstream target Elk, a transcription factor that promotes the expression of the c-Fos transcription factor. Jun expression is induced by the VAV1–Rac pathway(Terrell and Morrison 2019). Thus, the DAG–RAS pathway promotes the formation and activation of a dimeric complex, activator protein-1 (AP-1) composed of Jun/Fos, which plays a critical role in immune response, followed by IL-2 transcription. ERKs also phosphorylate the signal transducer and activator of transcription (STAT3) and LCK.
Negative Regulation of TCR signaling
Negative regulation of TCR signaling is also necessary to maintain the hyperactivation of the immune response associated with the pathway, which is accomplished through the involvement of several proteins and receptors.
SHP2-interacting transmembrane adaptor protein (SIT) is a transmembrane adaptor protein that interacts with the SH2-containing protein tyrosine phosphatase-2 (SHP2) via an ITIM, and the complex is important in the negative regulation of TCR-mediated signaling (Marie-Cardine, Kirchgessner et al. 1999). In resting human T-cells, phosphoprotein associated with glycosphingolipid microdomains (PAG) is tyrosine phosphorylated and associated with C-Src Tyrosine Kinase (CSK), an inhibitor of Scr-related protein tyrosine kinases. PAG overexpression, as a result, suppresses TCR-mediated signaling. In addition, the transmembrane protein cytotoxic T-lymphocyte antigen-4 (CLTA4) functions as a natural inhibitor. When TCR signaling needs to be regulated, ZAP-70 activates the process of active release and membrane translocation of CTLA4. At the membrane, CTLA4 interacts with SHP2, inhibiting TCR phosphorylation. Another way CTLA4 may inhibit T-cell function is by competing for the shared ligands B7-1 and B7-2 with CD28. CTLA4 is endocytosed when it is not needed to maintain a rapid T-cell activation response (Guntermann and Alexander 2002).
Relationship with Diseases
T cells play an important role in cell-mediated immunity, tumor cell defense, pathogenic organisms within cells, and rejection reactions. T-cell function dysregulation, whether due to a defect or an excess, has serious consequences for the organism, including immunodeficiency and autoimmunity. To elicit a T-cell response, only a few peptide-MHC complexes on APC are required. TCR is an extremely sensitive system.(Ahmad Mokhtar, Hashim et al. 2021). The TCR signaling pathway gives us a thorough understanding of how immune regulation defects cause immune-related diseases, as well as how the immune system can be better manipulated to overcome ailments like cancer, infection, and autoimmune diseases.
Conclusion
TCR signaling is thought to travel down from the TCR via the LAT to activate transcription factors required for T cell proliferation and effector function differentiation. Signaling appears to be branching sideways through numerous adaptor domains and integrating actin polymerization, which is likely to result in cytoskeletal changes. Signals are delivered to membrane-associated molecules like integrins via the recruitment of small GTPases to the plasma membrane. Integrin activation, in turn, affects the preservation of signaling-molecule-containing microclusters and, as a result, the extent of signal propagation. The regulation of these signaling cascades helps determine the activation threshold of naive T cells, ensuring that they are not initiated by self-peptide–MHC complexes but only respond to foreign peptide–MHC complexes. Effector T cells are more effective than naive T cells and, as a result, are more prone to self-reactivity and immune pathology. Negative regulators are upregulated in effector T cells to counteract this heightened sensitivity to activation and retain immune tolerance.
References
- Ahmad Mokhtar, A. M., et al. (2021). “The Role of RhoH in TCR Signalling and Its Involvement in Diseases.” Cells 10(4).
- Beach, D., et al. (2007). “Dual role of SLP-76 in mediating T cell receptor-induced activation of phospholipase C-gamma1.” J Biol Chem 282(5): 2937-2946.
- Gorentla, B. K. and X. P. Zhong (2012). “T cell Receptor Signal Transduction in T lymphocytes.” J Clin Cell Immunol 2012(Suppl 12): 5.
- Guirado, M., et al. (2002). “Phosphorylation of the N-terminal and C-terminal CD3-epsilon-ITAM tyrosines is differentially regulated in T cells.” Biochem Biophys Res Commun 291(3): 574-581.
- Guntermann, C. and D. R. Alexander (2002). “CTLA-4 suppresses proximal TCR signaling in resting human CD4(+) T cells by inhibiting ZAP-70 Tyr(319) phosphorylation: a potential role for tyrosine phosphatases.” J Immunol 168(9): 4420-4429.
- Koretzky, G. A. and N. J. Boerth (1999). “The role of adapter proteins in T cell activation.” Cell Mol Life Sci 56(11-12): 1048-1060.
- Krappmann, D., et al. (1999). “Molecular mechanisms of constitutive NF-kappaB/Rel activation in Hodgkin/Reed-Sternberg cells.” Oncogene 18(4): 943-953.
- Macian, F. (2005). “NFAT proteins: key regulators of T-cell development and function.” Nat Rev Immunol 5(6): 472-484.
- Marie-Cardine, A., et al. (1999). “SHP2-interacting transmembrane adaptor protein (SIT), a novel disulfide-linked dimer regulating human T cell activation.” J Exp Med 189(8): 1181-1194.
- Sewell, A. K. (2012). “Why must T cells be cross-reactive?” Nat Rev Immunol 12(9): 669-677.
- Terrell, E. M. and D. K. Morrison (2019). “Ras-Mediated Activation of the Raf Family Kinases.” Cold Spring Harb Perspect Med 9(1).
- van Boxel, G. I., et al. (2010). “An alternative conformation of the T-cell receptor alpha constant region.” J Mol Biol 400(4): 828-837.