Introduction
Solution /s are a cornerstone of almost all biological, biochemical, and molecular biology experiments, making it essential for every researcher to understand how to prepare, manipulate, and measure them accurately. The ability to make precise solutions can significantly impact the reliability of an experiment, as even small deviations in concentration or volume can compromise results and potentially invalidate months of work.
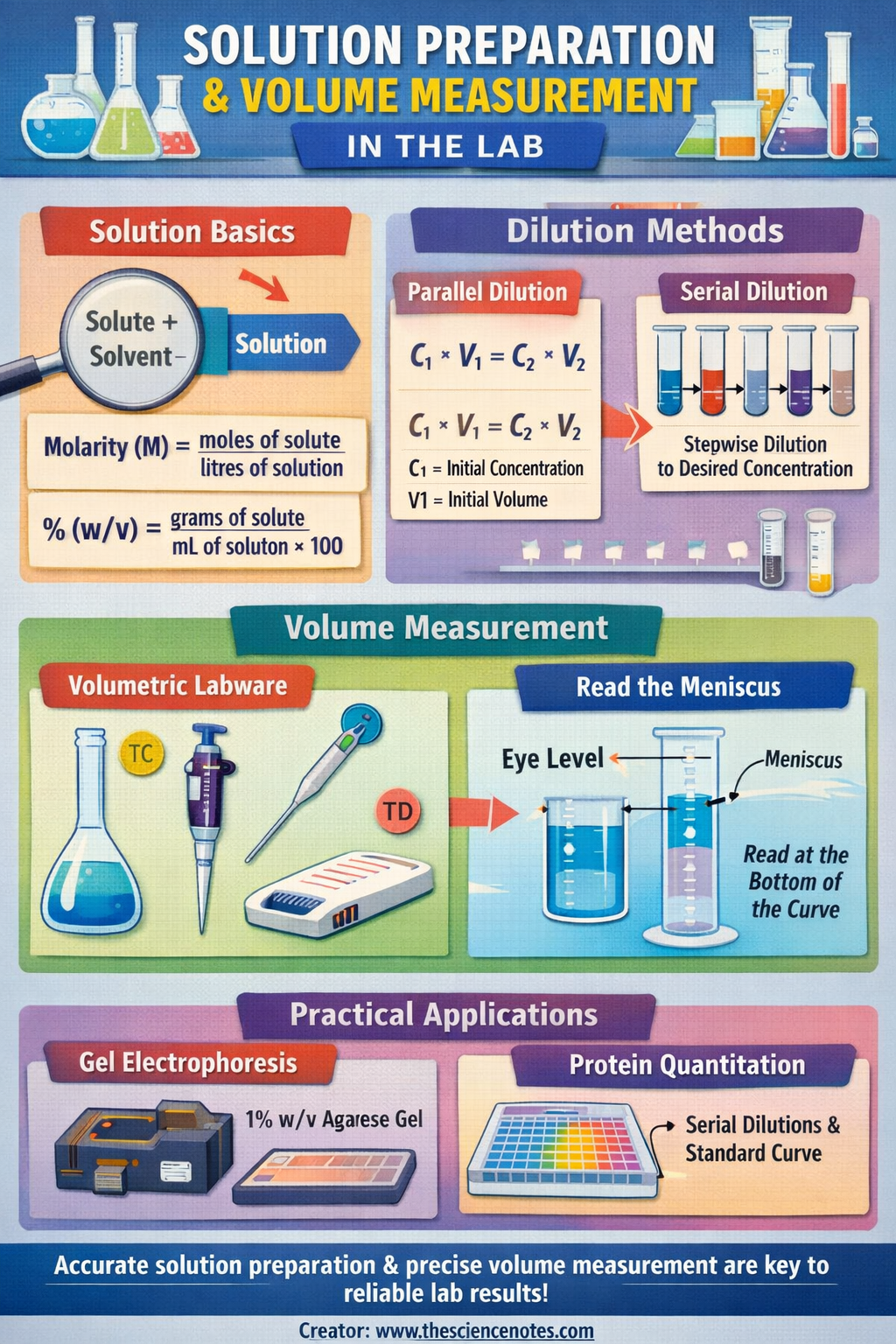
A solution is defined as a homogeneous mixture formed when a solute, the substance being dissolved, is combined with a solvent, the liquid medium that facilitates the dissolution. Proper solution preparation ensures uniform distribution of the solute at the molecular level, which is critical for reproducibility and experimental integrity.
This comprehensive guide will walk you through:
The fundamental components and types of solutions
Methods for calculating and preparing solution concentrations
Dilution techniques for achieving desired concentrations
Accurate volumetric measurement methods and labware selection
Practical applications in molecular biology and biochemistry experiments
Understanding the Composition of a Solution
A solution consists primarily of two components:
Solute: This is the substance that is being dissolved, which may exist as a solid, liquid, or gas. Examples include table salt, proteins, DNA, or chemical reagents. The solute is uniformly dispersed throughout the solvent when properly dissolved.
Solvent: This is the medium in which the solute dissolves, usually a liquid such as water, buffer, or an organic solvent. The solvent allows the solute molecules to interact and form a homogeneous mixture.
The proper ratio of solute to solvent is vital because the concentration of a solution determines the chemical environment for an experiment, influencing reactions, molecular interactions, and assay outcomes.
Quantifying Solution Concentration
Understanding concentration is crucial, as many experimental protocols require precise control over the amount of solute in a given volume of solvent.
1. Molarity (M)
Molarity is one of the most commonly used measures of concentration in the laboratory. It is defined as the number of moles of solute per liter of solution:
M = moles of solute / liters of solution
For example, a 1 M solution contains one mole of solute dissolved in one liter of total solution. Determining the number of moles requires knowledge of the solute’s molecular weight and the measured mass being used, highlighting the importance of accuracy in weighing chemicals.
2. Percent Concentration
Percent concentration is frequently used when expressing solutions as a fraction of solute relative to solvent.
a. Weight/Volume (% w/v)
This approach is commonly used for solid solutes:
% (w/v) = (grams of solute / mL of solution) × 100
Example: Preparing a 1% agarose gel for DNA electrophoresis requires 1 gram of agarose dissolved in 100 mL of buffer solution.
b. Volume/Volume (% v/v)
Used when both solute and solvent are liquids:
% (v/v) = (volume of solute / volume of solution) × 100
This method is commonly applied in preparing alcohol solutions or other liquid chemical mixtures.
Stock Solutions and the Importance of Dilution
Stock Solutions
They (stock solutions) are highly concentrated solutions of stable compounds that are stored for repeated use. They are often labeled as multiples, such as 10X or 50X, indicating how many times more concentrated they are compared to the working solution.
Example: A 10X stock buffer can be diluted tenfold to prepare the working 1X solution.
Advantages: Saves preparation time, ensures consistency across experiments, and reduces the risk of measurement errors when large volumes are required.
Dilution Techniques
Dilution reduces the concentration of a solution by adding additional solvent. Accurate dilution is critical for achieving desired experimental conditions.
Basic Dilution Formula:
C1 × V1 = C2 × V2
Where:
C1 = concentration of stock solution
V1 = volume of stock solution needed
C2 = desired final concentration
V2 = final total volume
This formula allows researchers to calculate precisely how much stock solution and solvent are required to achieve the target concentration.
Parallel (Direct) Dilution
Parallel dilution involves preparing the desired final concentration in a single step from a stock solution.
Example: Diluting a 10X buffer stock to a 1X working solution by combining 1 part stock with 9 parts solvent.
Limitation: When the required volume of stock solution is very small, measurement errors can become significant, making the process less accurate for extremely low concentrations.
Serial Dilution
Serial dilution overcomes the limitations of parallel dilution by incrementally diluting a solution in multiple steps:
Step 1: Dilute the stock solution to an intermediate concentration
Step 2: Use the intermediate solution to prepare an even lower concentration
Repeat as necessary until the target concentration is reached
Particularly, serial dilutions are useful in preparing standard curves, which require a range of known concentrations to quantify unknown samples, such as proteins or nucleic acids.
Measuring Volumes Accurately in the Laboratory
Accurate volume measurement is essential for precise solution preparation. Laboratory containers vary in design and accuracy, so choosing the appropriate labware is critical.
Non-Volumetric Containers
Examples: Beakers, Erlenmeyer flasks
Purpose: Mixing, storage, and rough volume approximations
Limitations: Graduations are approximate, not suitable for experiments requiring exact concentrations
Volumetric Labware
Designed for high-precision measurements, volumetric labware includes:
TC (To Contain): Holds an exact volume, found on volumetric flasks and some graduated cylinders
TD (To Deliver): Designed to dispense a precise volume, found on pipettes and syringes
Selecting the smallest suitable volumetric device improves measurement accuracy.
Common Volumetric Instruments
Volumetric Flasks: Used to prepare solutions of precise concentrations. After dissolving the solute, solvent is added until reaching the calibration mark, a process called Q.S. (quantity sufficient).
Graduated Cylinders: Suitable for measuring volumes above 50 mL; moderately accurate.
Serological Pipettes: Measure and deliver 0.1–50 mL of liquid.
Micropipettes: Measure small volumes from 0.2 µL to 5 mL with high precision.
Hamilton Syringes: Glass syringes used for microliter volumes when plastic tips are incompatible.
Reading the Meniscus
The meniscus is the curved surface of a liquid in a container caused by surface tension.
In aqueous solutions, the meniscus is concave
Always read the lowest point at eye level to ensure accuracy
Applications in Laboratory Experiments
1. Gel Electrophoresis
Gel electrophoresis separates DNA fragments by size using an agarose matrix:
Requires preparation of percent weight/volume agarose solutions (e.g., 1% w/v agarose)
Running buffers are often diluted from concentrated 10X stock solutions
Example: To make 1X running buffer, mix 1 volume of 10X stock with 9 volumes of purified water
2. Microplate Reader Experiments
Microplate reader assays rely on precise solution preparation:
Unknown protein concentrations are determined using standards prepared from serial dilutions
A standard curve is generated by plotting known concentrations against measured values
Serial dilution ensures evenly spaced standards and reduces pipetting errors
Key Takeaways
Accurate solution preparation is essential for reliable and reproducible experiments
Concentration can be expressed as molarity, % w/v, or % v/v, depending on the solute and solvent
Stock solutions simplify repeated experiments but must be accurately diluted
Serial and parallel dilutions are fundamental techniques for achieving target concentrations
Selecting appropriate volumetric labware and using correct techniques ensures precision
Reading the meniscus and using consistent pipetting techniques prevents measurement errors
Remember: Accuracy and precision are crucial in all aspects of solution preparation, whether preparing buffers, gels, or standards for assays.