Liver organoids have become an important tool for studying human liver biology, disease mechanisms, drug responses, inflammation, and regenerative medicine. Traditional two-dimensional liver cell cultures can provide useful information, but they often fail to reproduce the complex cellular communication that occurs inside native liver tissue. The liver is not made of hepatocytes alone. It contains epithelial, endothelial, mesenchymal, and immune cell populations that interact continuously to regulate metabolism, immune defense, injury response, and tissue repair.
Among these cell types, Kupffer cells are especially important. Kupffer cells are liver-resident macrophages. They help detect pathogens, clear debris, process antigens, regulate inflammation, and participate in liver repair and regeneration. Because of these functions, they are central to liver pathogenesis in conditions such as sepsis-related liver dysfunction, inflammatory liver injury, metabolic liver disease, fibrosis, and infection-associated hepatic stress.
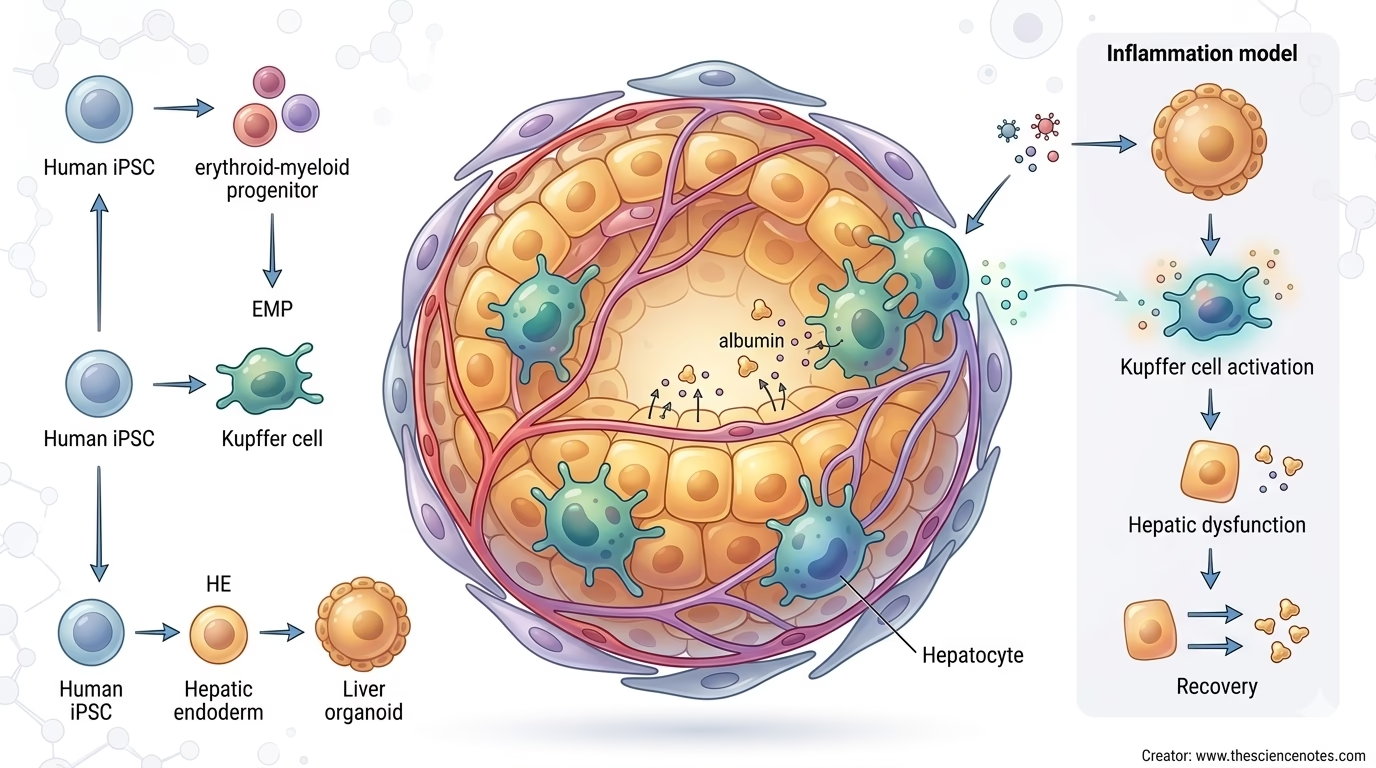
A recent STAR Protocols article by Yang Li, Yunzhong Nie, and Hideki Taniguchi describes a protocol for generating human induced pluripotent stem cell-derived liver organoids containing Kupffer cells, also called KuLOs. The workflow integrates Kupffer cell progenitors into liver organoids by generating multiple cell lineages from human iPSCs and then combining them in a scaffold-free organoid co-culture system.
What Are Liver Organoids Containing Kupffer Cells?
Liver organoids containing Kupffer cells are three-dimensional liver-like structures that include both hepatic and immune components. In the protocol described by Li and colleagues, the organoids are generated from human iPSCs and contain multiple iPSC-derived lineages:
| Cell Component | Role in the Organoid Model |
|---|---|
| Hepatic endoderm | Gives rise to liver epithelial or hepatocyte-like components |
| Endothelial cells | Support vascular-like signaling and tissue organization |
| Mesenchymal cells | Provide stromal support and developmental cues |
| Erythroid-myeloid progenitors | Serve as Kupffer cell progenitors |
| Kupffer-like macrophages | Provide liver-resident immune functions |
The resulting organoids are referred to as KuLOs, meaning Kupffer cell-containing liver organoids.
The key advantage of this model is that it does not treat the liver as a purely epithelial organ. Instead, it incorporates tissue-resident macrophage biology into the organoid system. This makes the model more relevant for studying liver inflammation, immune-mediated hepatic dysfunction, and recovery after inflammatory injury.
Why Kupffer Cells Matter in Liver Organoid Models
Kupffer cells are among the most abundant resident macrophage populations in the human body. They are strategically positioned in the liver to monitor blood entering from the portal vein and hepatic artery. This exposes them to nutrients, microbial products, toxins, immune signals, and circulating debris.
Their functions include:
- Detecting inflammatory and microbial signals
- Clearing apoptotic cells and damaged material
- Producing cytokines and chemokines
- Supporting liver repair and regeneration
- Participating in iron handling and antigen processing
- Modulating hepatocyte function during inflammation
- Contributing to liver injury when immune activation becomes excessive
Because Kupffer cells can either protect the liver or contribute to injury depending on context, including them in organoid models is valuable. A hepatocyte-only organoid may show metabolic activity, but it cannot fully model immune-driven hepatic dysfunction. A Kupffer cell-containing organoid can better represent how immune activation affects liver function.
This is particularly important for modeling sepsis-associated liver dysfunction, where systemic inflammation and endotoxin exposure can impair hepatic function. In the KuLO system, endotoxin stimulation can activate Kupffer cells, trigger inflammatory cytokine release, and reduce hepatic performance. This provides a controlled human-cell platform for studying inflammation-related liver injury and recovery.
Why Use Human iPSCs for Generating KuLOs?
Human induced pluripotent stem cells are useful because they can be expanded and differentiated into many cell types. In organoid research, iPSCs provide a flexible starting material for creating complex, multicellular models.
Using human iPSCs offers several advantages:
1. Human Biological Relevance
Animal models are valuable but do not always reproduce human liver development, immune signaling, drug metabolism, or inflammatory responses. Human iPSC-derived models can help bridge this gap.
2. Multilineage Differentiation
The KuLO protocol uses iPSCs to generate several cell lineages needed for organoid assembly, including hepatic endoderm, endothelial cells, mesenchymal cells, and erythroid-myeloid progenitors.
3. Reproducibility and Scalability
iPSC-derived systems can be expanded and standardized more effectively than many primary human tissue systems, although line-to-line variability still needs to be controlled.
4. Disease Modeling Potential
Patient-specific or genetically edited iPSC lines may be used to study inherited liver disorders, immune dysregulation, inflammatory susceptibility, or disease-associated variants.
5. Drug Testing and Therapeutic Screening
Because KuLOs combine hepatic and immune elements, they may support testing of compounds that modulate inflammation, hepatocyte function, macrophage activation, or liver recovery.
Overview of the KuLO Generation Strategy
The protocol described by Li and colleagues follows a developmental logic. Instead of simply adding mature macrophages to preformed organoids, it generates erythroid-myeloid progenitors, or EMPs, from human iPSCs and incorporates them into liver organoids during co-culture.
This matters because Kupffer cells arise developmentally from early myeloid progenitor populations. By integrating EMPs into the organoid system, the protocol aims to mimic aspects of tissue-resident macrophage development and integration.
The general workflow can be summarized in four major phases:
- Prepare and expand high-quality human iPSCs
- Differentiate iPSCs into EMPs and hepatic endoderm
- Combine EMPs with hepatic, endothelial, and mesenchymal lineages
- Allow organoids to mature and characterize Kupffer cell integration
The final KuLOs contain multiple cell types and can be evaluated for hepatic maturation, Kupffer cell differentiation, immune activation, and functional recovery after inflammatory challenge.
Phase 1: Preparing Human iPSCs for Differentiation
Successful KuLO generation starts with high-quality human iPSC culture. The protocol emphasizes that iPSCs should be healthy, maintained under appropriate culture conditions, and passaged before differentiation.
This step is critical because poor iPSC quality can reduce differentiation efficiency across multiple lineages. If iPSCs are stressed, overgrown, spontaneously differentiated, or kept at excessive passage numbers, downstream hepatic and hematopoietic differentiation may be compromised.
Important quality considerations include:
- Maintaining iPSC colonies in an undifferentiated state
- Avoiding spontaneous differentiation before starting the protocol
- Using appropriate extracellular matrix coating
- Controlling passage timing
- Using iPSCs with preserved differentiation potential
- Coordinating separate differentiation timelines for different cell lineages
The protocol notes that hepatic endoderm and EMPs should not be cryopreserved for co-culture in this workflow. Instead, they are recommended for fresh use, while endothelial and mesenchymal progenitors may be handled according to previously established procedures.
This scheduling detail matters. Organoid assembly depends on having multiple cell lineages ready at the same time.
Phase 2: Generating Erythroid-Myeloid Progenitors from Human iPSCs
One of the central innovations of the protocol is the efficient generation of erythroid-myeloid progenitors, or EMPs. These progenitors are used as the source of Kupffer-like cells in the organoid system.
EMPs are important because they represent an early hematopoietic population with myeloid potential. In the KuLO workflow, they are differentiated from human iPSCs through a sequence designed to promote hemogenic and myeloid lineage specification.
During the differentiation process, researchers monitor the emergence of relevant progenitor markers. The protocol highlights markers such as:
- CD43
- CD34
- CD115, also known as CSF-1R
The target EMP population is described as CD43-positive, CD34-positive, and CD115-positive. These markers help confirm that the differentiation process is producing a suitable Kupffer cell progenitor population.
Flow cytometry is used to examine differentiation efficiency. This is an important quality-control checkpoint because the success of Kupffer cell integration depends on the quality and identity of the EMP population.
If EMP differentiation is poor, the final organoids may contain fewer Kupffer-like cells or show inconsistent immune behavior. The protocol recommends monitoring differentiation carefully and optimizing iPSC seeding density and reagent quality when yields are low.
Phase 3: Generating Hepatic Endoderm from Human iPSCs
The hepatic component of the organoid begins with differentiation of human iPSCs into hepatic endoderm, often abbreviated as HE. Hepatic endoderm represents a developmental stage on the path toward hepatocyte-like cells.
The HE differentiation process includes two broad stages:
- Definitive endoderm specification
- Hepatic endoderm differentiation
This reflects normal developmental biology. During embryonic development, liver tissue arises from definitive endoderm, which then receives signals that drive hepatic specification.
In the KuLO protocol, successful HE differentiation is assessed using morphology, flow cytometry, and gene expression analysis. By the end of the HE differentiation phase, the cells are expected to show hepatic endoderm characteristics, including polygonal morphology and expression of relevant markers.
Important HE markers include:
- CD326, also known as EpCAM
- CD133
- HNF4A
- AFP
The protocol also notes that definitive endoderm markers such as CXCR4 and CER1 are expected to show transient expression during differentiation, while pluripotency genes such as NANOG and POU5F1 should decrease.
A strong HE population is essential because it forms the hepatic foundation of the organoid. Poor HE differentiation can lead to failure of organoid formation, reduced hepatic function, or poor maturation.
Phase 4: Co-Culturing Multiple iPSC-Derived Lineages to Generate KuLOs
After generating the required cell types, the protocol combines them in a scaffold-free co-culture system. The organoids are formed in U-bottom microwell plates, which help cells self-aggregate into three-dimensional structures.
The main cell populations used for KuLO generation are:
- iPSC-derived hepatic endoderm
- iPSC-derived endothelial cells
- iPSC-derived mesenchymal cells
- iPSC-derived erythroid-myeloid progenitors
The protocol recommends a lineage ratio in which hepatic endoderm is the dominant population, with endothelial, mesenchymal, and EMP populations included at lower but meaningful proportions. The provided ratio is 5:1:1:1 for HE, ECs, MCs, and EMPs respectively.
This co-culture design supports organoid formation while enabling EMP-derived macrophage development in and around the organoids. During co-culture, EMPs expand and differentiate toward a Kupffer-like phenotype. Meanwhile, the liver organoids undergo functional maturation.
The model is designed so that organoids gradually develop hepatic features while also incorporating immune-cell functionality.
What Are KuLOs Expected to Contain?
KuLOs are multicellular structures. They are not composed of a single liver-like cell type. Their value comes from the interaction of several lineages.
Hepatic Cells
The hepatic component contributes liver-related functions, including albumin secretion, expression of hepatic markers, and metabolic activity.
Endothelial Cells
Endothelial cells help represent vascular-associated signaling. Although the protocol notes that the endothelial network remains limited compared with native liver tissue, this cell type adds important tissue complexity.
Mesenchymal Cells
Mesenchymal cells provide stromal support and developmental cues. They may help organize organoid architecture and influence hepatic maturation.
Kupffer-Like Cells
The EMP-derived macrophage population differentiates toward a Kupffer-like phenotype, expressing markers such as:
- CD45
- CD14
- CD163
By approximately two weeks of co-culture, the protocol reports that most EMP-derived cells can express Kupffer-associated markers. These Kupffer-like cells contribute immune responsiveness and help model liver inflammation.
Characterizing Kupffer Cells in Liver Organoids
Characterization is a major part of the KuLO protocol. Generating organoids is not enough; researchers must confirm that the desired cell populations are present and functional.
Several approaches are used.
Flow Cytometry
Flow cytometry helps analyze hematopoietic and Kupffer-like cell differentiation. The protocol recommends examining EMP-derived cells during co-culture and assessing Kupffer-associated markers such as CD45, CD14, and CD163.
Flow cytometry can also help distinguish other organoid cell populations using lineage markers such as:
- CD326 for hepatic cells
- CD31 for endothelial cells
- CD45 for hematopoietic cells
However, the protocol notes that dissociation of organoids may not perfectly represent the actual cellular composition because some cell clusters may remain undissociated.
Whole-Mount Immunostaining
Whole-mount immunostaining allows researchers to visualize organoids in three dimensions. This can show the spatial organization of hepatic cells and Kupffer-like cells within or around the organoid structures.
Markers such as hepatic proteins and macrophage markers can be used to confirm multilineage organization.
Hepatic Function Assays
The organoids can be assessed for liver-like function through readouts such as:
- Albumin secretion
- Hepatic marker gene expression
- CYP3A4 enzyme activity
- Ammonia metabolism
Albumin secretion is especially useful because it provides a functional indicator of hepatic maturation over time. The protocol reports that albumin secretion increases during co-culture and stabilizes as KuLOs mature.
Gene Expression Analysis
Gene expression can be used to assess hepatic maturation, Kupffer cell identity, inflammatory activation, and lineage-specific development. This is particularly useful for comparing KuLOs with organoids lacking Kupffer cells or with primary liver tissue references.
Expected Outcomes of KuLO Generation
The expected outcome is a functional human iPSC-derived liver organoid system containing Kupffer-like cells. These organoids include progenitors and differentiated cells from multiple lineages and can model immune-hepatic interactions.
By approximately day 14 of co-culture, KuLOs are expected to reach a more mature state. At this stage, hepatic functions stabilize and EMP-derived cells show Kupffer cell marker expression.
Reported functional features include:
- Increased albumin secretion over time
- Upregulation of hepatic markers such as ALB, A1AT, and CPS1
- CYP3A4 activity
- Ammonia metabolism
- Differentiation of EMPs into Kupffer-like cells
- Enrichment of Kupffer-related functions such as antigen processing and iron ion homeostasis
- Responsiveness to endotoxin-induced inflammatory stimulation
These outcomes make KuLOs a useful platform for studying how liver-resident macrophages influence hepatic function during inflammation.
Modeling Sepsis-Related Liver Dysfunction
One of the most important applications of KuLOs is modeling inflammation-associated liver dysfunction, including conditions related to sepsis.
Sepsis is characterized by systemic inflammation, immune dysregulation, and organ dysfunction. The liver is heavily involved because it filters blood, responds to inflammatory mediators, produces acute-phase proteins, and regulates metabolism during systemic stress.
In the KuLO model, endotoxin stimulation can activate Kupffer-like cells. This can lead to:
- Kupffer cell activation
- Pro-inflammatory cytokine release
- Impaired hepatic function
- Reduced liver organoid performance
- Recovery after removal of inflammatory stimuli
This makes KuLOs useful for studying both injury and recovery. Researchers can examine how inflammatory signals disrupt hepatocyte-like function and how tissue function may be restored after inflammatory mediators are removed.
This type of model may be valuable for therapeutic screening. Compounds that reduce harmful inflammation or promote hepatic recovery could be evaluated in a human multicellular organoid context.
Research Applications of Human iPSC-Derived KuLOs
1. Liver Inflammation Research
KuLOs provide a platform to study how Kupffer cells regulate inflammatory signaling inside liver-like tissue. This is useful for exploring cytokine networks, macrophage activation states, and hepatocyte responses to immune stress.
2. Sepsis and Acute Liver Dysfunction
Because KuLOs can respond to endotoxin exposure, they are useful for modeling sepsis-like inflammatory liver dysfunction. Researchers can study how immune activation impairs hepatic function and how recovery occurs.
3. Liver Regeneration and Repair
Kupffer cells participate in liver repair after injury. KuLOs may help researchers investigate macrophage-derived signals that support or inhibit liver regeneration.
4. Drug-Induced Liver Injury
Many drug-induced liver injury models focus primarily on hepatocytes. Adding Kupffer-like cells may help model immune-mediated liver toxicity or inflammatory amplification of drug responses.
5. Metabolic Liver Disease
Chronic liver diseases such as fatty liver disease involve interactions between hepatocytes, macrophages, endothelial cells, and stromal cells. KuLOs may support mechanistic studies of inflammatory-metabolic crosstalk.
6. Fibrosis Research
Although KuLOs are not a complete fibrosis model, the inclusion of mesenchymal cells and Kupffer-like cells may support future adaptation for studying fibrogenic signaling and macrophage-stromal interactions.
7. Personalized Medicine
Patient-specific iPSCs could theoretically be used to generate KuLOs for studying individual susceptibility to liver inflammation, immune dysregulation, or genetic liver disease.
8. Therapeutic Screening
KuLOs may provide a platform for testing anti-inflammatory agents, hepatoprotective compounds, macrophage-modulating drugs, or regenerative therapies.
Why This Protocol Is Innovative
The main innovation is the integration of Kupffer cell progenitors into human iPSC-derived liver organoids through a developmental strategy.
Many organoid systems lack resident immune cells. This limits their ability to model inflammation and immune-tissue communication. Other methods may add macrophages externally, but these approaches may not fully replicate tissue-resident macrophage integration.
The KuLO protocol addresses this by generating EMPs from iPSCs and co-culturing them with hepatic and supportive lineages. This allows Kupffer-like cells to emerge within a developing organoid environment.
The result is a more complex liver model that better reflects the multicellular nature of human liver tissue.
Quality Control Considerations
Reliable KuLO generation depends on several checkpoints.
iPSC Quality
Healthy, undifferentiated iPSCs are essential. Poor iPSC culture quality can reduce both hepatic and hematopoietic differentiation.
EMP Differentiation
The EMP population should be assessed using markers such as CD43, CD34, and CD115. Low EMP yield may reduce Kupffer cell formation.
HE Differentiation
Hepatic endoderm should show appropriate morphology and marker expression. Low HE quality can impair organoid formation and hepatic maturation.
Organoid Formation
Cells should aggregate into spheroid-like structures after seeding into microwells. Failure to form organoids may indicate poor HE viability, inadequate cell preparation, or missing survival-supporting conditions.
Kupffer Cell Marker Expression
Kupffer-like differentiation should be monitored using markers such as CD45, CD14, and CD163.
Hepatic Function
Albumin secretion, CYP activity, ammonia metabolism, and hepatic gene expression can help assess organoid maturation.
Limitations of KuLO Models
Although KuLOs are an important advancement, they are not perfect replicas of adult human liver tissue.
Limited Hepatocyte Maturity
The protocol notes that KuLOs show hepatic function but remain less mature than primary human hepatocytes. This is a common limitation of many iPSC-derived hepatic systems.
Incomplete Endothelial Network
The organoids contain endothelial cells, but they do not fully reproduce the complex vascular architecture of the liver. This may limit applications focused on sinusoidal biology or vascular inflammation.
Organoid Variability
Organoid size, cell composition, and maturation may vary across wells, iPSC lines, and differentiation batches.
Technical Complexity
The workflow requires coordination of multiple cell lineages with different differentiation timelines. This increases experimental complexity compared with simpler organoid models.
Partial Immune Representation
KuLOs include Kupffer-like macrophages, but the liver immune environment also contains other immune populations, such as lymphocytes, neutrophils, dendritic cells, and circulating monocytes. These are not fully represented.
Troubleshooting Concepts
The protocol identifies several common problems and practical causes.
Low EMP Differentiation Efficiency
Low EMP yield may result from poor iPSC quality, suboptimal starting density, or degraded cytokines and growth factors. The protocol recommends optimizing iPSC seeding density and using fresh or properly stored supplements.
Low HE Differentiation Efficiency
Poor HE differentiation can occur when iPSCs have reduced differentiation potential, when seeding density is not optimal, or when growth factors lose activity. Marker analysis and morphology checks are important for diagnosing this problem.
Failure of Organoid Formation
If cells remain scattered instead of clustering, likely causes include poor HE differentiation, low HE viability, or insufficient support during initial co-culture. The protocol emphasizes the importance of cell viability and appropriate co-culture setup.
Organoid Loss During Medium Changes
Because organoids are not physically attached to the microwells, careless handling can disturb or aspirate them. Gentle and consistent medium exchange is important.
Inconsistent Kupffer Cell Differentiation
This may reflect variability in EMP quality, co-culture conditions, or timing. Flow cytometry at multiple time points can help track differentiation dynamics.
Frequently Asked Questions
What are KuLOs?
KuLOs are human iPSC-derived liver organoids containing Kupffer-like cells. They combine hepatic, endothelial, mesenchymal, and hematopoietic-derived immune components in a three-dimensional organoid model.
What are Kupffer cells?
Kupffer cells are liver-resident macrophages. They help regulate inflammation, clear debris, process antigens, participate in iron metabolism, and support liver repair.
Why include Kupffer cells in liver organoids?
Including Kupffer cells allows organoids to model immune-hepatic interactions. This is important for studying liver inflammation, sepsis-related dysfunction, drug-induced injury, and tissue repair.
What are EMPs?
EMPs are erythroid-myeloid progenitors. In this protocol, they are generated from human iPSCs and used as Kupffer cell progenitors.
What markers are used to identify EMPs?
The protocol highlights CD43, CD34, and CD115 as important EMP-associated markers.
What markers are used to identify Kupffer-like cells?
Kupffer-like cells in KuLOs are characterized using markers such as CD45, CD14, and CD163.
What markers are used to evaluate hepatic endoderm?
Hepatic endoderm can be assessed using markers such as EpCAM/CD326, CD133, HNF4A, and AFP, along with downregulation of pluripotency markers.
How long does KuLO maturation take?
The co-culture phase takes about two weeks. Around day 14, KuLOs are expected to show more mature hepatic function and differentiated Kupffer-like cells.
What liver functions can KuLOs model?
KuLOs can show albumin secretion, hepatic marker expression, CYP3A4 activity, ammonia metabolism, and inflammatory response to endotoxin stimulation.
Are KuLOs equivalent to adult human liver tissue?
No. KuLOs are advanced in vitro models, but they remain less mature and less structurally complex than native adult human liver.
Conclusion
Human iPSC-derived liver organoids containing Kupffer cells represent an important step toward more realistic in vitro liver models. By integrating liver-resident macrophage-like cells into a multicellular organoid system, KuLOs allow researchers to study immune-hepatic communication in a controlled human-cell platform.
The protocol by Li, Nie, and Taniguchi focuses on generating erythroid-myeloid progenitors and hepatic endoderm from human iPSCs, then combining these cells with iPSC-derived endothelial and mesenchymal lineages to establish Kupffer cell-containing liver organoids. The resulting organoids support hepatic maturation, Kupffer-like cell differentiation, inflammatory activation, and recovery after inflammatory challenge.
For liver disease research, this model is especially valuable because many liver conditions involve communication between hepatocytes and immune cells. Sepsis-related liver dysfunction, inflammatory injury, drug toxicity, regeneration, and chronic disease progression all involve macrophage-hepatic interactions. KuLOs provide a practical research platform for studying these processes in a human iPSC-derived system.
Although limitations remain, including incomplete hepatocyte maturity and limited endothelial network formation, KuLOs offer a more complex and biologically relevant model than hepatocyte-only organoids. As organoid technology advances, Kupffer cell-containing liver organoids may become increasingly useful for disease modeling, drug screening, regenerative medicine, and precision hepatology.
Source Attribution
This article is based on the scientific content provided from:
Li, Y., Nie, Y., & Taniguchi, H. “Protocol for generating liver organoids containing Kupffer cells using human iPSCs.” STAR Protocols, Volume 6, Issue 4, 2025.
Related referenced work:
Li, Y. et al. “Integration of Kupffer cells into human iPSC-derived liver organoids for modeling liver dysfunction in sepsis.” Cell Reports, 2024.