Bacterial transformation is the process of transfer of foreign genetic materials from the external environment through the cell membrane.
It’s a horizontal form of gene transfer first discovered in Streptococcus pneumoniae by British bacteriologist Frederick Griffith in 1928. He tested whether or not heat-killed bacteria might be used to vaccinate mice against pneumonia and found that a non-virulent strain of Streptococcus pneumoniae could be rendered virulent by exposing it to heat-killed virulent strains. He theorized the transforming principle based on this experiment, claiming that the heat-killed strain was responsible for making the innocuous strain infectious. Morton Mandel and Akiko Higo found in 1970 that E. coli may take up DNA from bacteriophage.
Even when a donor cell is not living, the gene transfer process in transformation necessitates the presence of persistent DNA in the environment. Some bacterial genera that have been exposed to extreme and harsh environmental conditions leak DNA into the environment. The bacteria’s major goal is to change its ability to take in extracellular genetic material from the environment. Competent cells are bacteria that have this ability. These capable cells are also able to adapt to changes in the environment and acquire genes through the natural transformation process.
But not all competent bacterial cells are capable of taking up DNA from the environment. It needs to be artificially competent with the use of chemicals or electric pulses. Chemicals like calcium phosphate are used with heat shock treatment that causes the DNA to enter into the cell. Electric pulse methods like electroporation make the bacterial cell-permeable that allows DNA to enter into the cell.
Factors that affect transformation efficiency
- The concentration of DNA (<10ng of DNA is used for efficacy).
- Supercoiled DNA is efficient than linear DNA
- The high concentration of salt lowers the transformation efficiency during electroporation.
- Ligase must be heat-inactivated as it inhibits the electroporation of cells.
- The time between transformation and plating.
- SOB medium is most suitable for transformation.
- Freezing and thawing of the cell decrease transformation efficiency.
Four steps are involved during bacterial transformation
- Preparation of competent cells
- Transformation
- Cell recovery period
- Cell plating
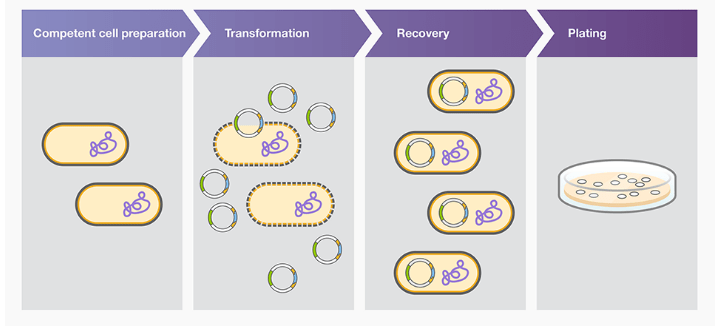
Preparation of competent cell
- The competent cells must be prepared and E. coli is the most commonly used bacterial species.
- The competency of E. coli is very low, therefore; the use of chemicals or electroporation must be administered to the cell to make it competent.
- Use of chemicals like calcium phosphate or calcium chloride is added with the cell and chilled to make the cell-permeable and it is incubated with external DNA.
- Heat shock treatment is given at 42°C for 60 to 120 seconds which opens up the cell membrane and causes DNA to enter into the cell.
- Heat shock treatment must be given when the cells are in the log phase of the growth.
- During electroporation, the harvested cells are washed with chilling de-ionized water repeatedly to remove salts and other components. Then the salts are finally pelleted and suspended in 10% glycerol.
Transformation
- Bacterial transformation is carried out in two ways: Chemical transformation and electroporation of competent cells.
- The choice of method depends on the transformation efficiency and available resources.
- Competent cells should be thawed on ice and cells should be gently mixed by shaking and tapping before the transformation.
- Heat shock treatment is also performed at 37 to 42°C for 25 to 45 seconds,
- The heat-shocked cells are placed on the ice for less than 2 minutes before performing the next step.
- Electroporation of cells is performed by using an electroporator where competent cells and DNA are exposed to a high voltage electric field which opens up the pores of the cell membrane and DNA enters into the cell.
Cell recovery period
- The cells after heat shocked or electroporation is cultured in an antibiotic-free liquid medium to allow expression of antibiotic genes for a short period of time.
- For the cell recovery, the transformed cells are cultured in 1ml of SOC medium and incubated at 37°C.
- SOC medium contains glucose and MgCl2 which increases the transformation efficiency.
Cell plating
- The cells are plated on LB agar along with antibiotics and other agents for identification and recovery of transformants.
- If blue or white screening is performed, X-Gal and IPTG must be included in the agar plate.
- The plates must be pre-warmed before cell plating to maintain a favorable growth temperature and to prevent contamination and mixed colonies.
- After cell plating, it must produce a sufficient amount of individual, distinct colonies for further screening.
Calculation of transformation efficiency

The colonies need to be screened further to detect the presence of desired plasmid or not. After conformation, the colonies are employed in downstream applications such as plasmid isolation, sub-cloning, transfection and protein expression.
References:
- https://www.sigmaaldrich.com/technical-documents/protocols/biology/transformation.html#:~:text=Bacterial%20transformation%20is%20a%20process,Avery%20et%20al%20in%201944.
- https://www.addgene.org/protocols/bacterial-transformation/
- https://www.khanacademy.org/science/biology/biotech-dna-technology/dna-cloning-tutorial/a/bacterial-transformation-selection
- https://microbenotes.com/bacterial-transformation/
- https://www.thermofisher.com/np/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/molecular-cloning/transformation/bacterial-transformation-workflow.html