Ethanol, also known as ethyl alcohol or EtOH, is a clear and colorless liquid known for its pleasant odor. It’s a key component in alcoholic beverages like beer and wine, which are responsible for intoxication. This article explores the industrial production of ethanol and its various applications.
Ethanol has a slightly sweet taste when diluted in water but burns in more concentrated solutions.
Production of Ethanol
Large industrial plants are the primary sources of ethanol production, although some individuals produce their own ethanol. For over a century, ethanol has been produced from various agricultural products, including those rich in starch, sugar, and cellulose.
Ethanol can be made from three main types of materials:
- Sugar Sources: Includes beet, sugar cane, sweet sorghum, and fruits.
- Starchy Materials: Such as corn, milo, wheat, rice, potatoes, cassava, and sweet potatoes.
- Cellulose Materials: Like wood, used paper, and crop residues.
Uses of Ethanol
Ethanol serves various purposes:
- Chemical Feedstock: Due to its reactivity, ethanol is crucial in the chemical industry.
- Solvent: Used in industries for colors, oils, waxes, explosives, cosmetics, etc.
- General Utility: Functions as a disinfectant, cleaning agent, and illumination source.
- Fuel: Blended with gasoline, it’s known as gasohol and used in automobiles.
Sorbitol: Biosynthesis, Degradation Pathway, and Applications (thesciencenotes.com)
Microbial Production of Ethanol
Ethanol production using microbial processes has become increasingly prevalent, utilizing organic feedstocks and plant substances, such as molasses, in the production process of ethanol. This section outlines the key steps involved in microbial production.
Historical Perspective
Historically, ethanol was primarily produced through fermentation processes. However, for many years, an alternative method involved the chemical synthesis of ethanol via the catalytic hydration of ethylene.
Modern Approach
Recent research has emphasized the use of microbial fermentation for ethanol production, both as a chemical and fuel. This method has gained popularity due to its sustainability and eco-friendliness.
1. Feedstock Selection
In contemporary ethanol production, various feedstocks are employed, including sugar beet, potatoes, corn, cassava, and sugar cane. These feedstocks serve as the basis for ethanol production.
2. Medium Preparation
The manufacturing process begins with the preparation of the fermentation medium. Three primary categories of substrates are utilized:
- Starch-containing substrates
- Sugarcane juice, molasses, sugar beet juice
- Wood waste or processed wood
It is important to note that the hydrolysis of starch is required before yeast strains are introduced into the medium. This hydrolysis, often accompanied by cellulose enzymes of microbial origin, yields reducing sugars necessary for the ethanol production process.
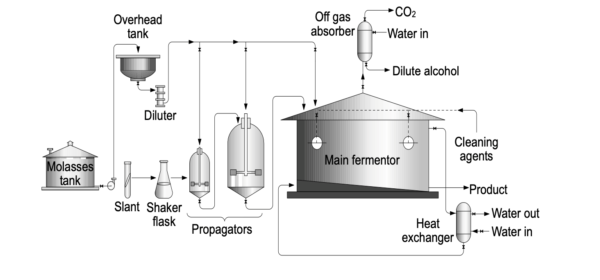
3. Fermentation Process
Fermentation is a critical phase in ethanol production, and it occurs continuously within massive fermenters. The specific methods employed may vary from one nation to another, with countries like India, Brazil, Germany, and Denmark each having their unique alcohol production technologies.
Despite cultural variations, the fermentation conditions typically involve maintaining a pH of 5 and a temperature of 35°C. While conventional fermentation methods often take several days to complete, ethanol production begins within just 12 hours.
Following the fermentation process’s completion, cells are separated to obtain yeast biomass, which can be repurposed for the production of single-cell proteins (SCPs) for animal feed. Additional processing is undertaken to recover ethanol from the culture medium or supernatant.
Continuous cell recycling during fermentation has been adopted to expedite the fermentation period, making the process more efficient.
4. Ethanol Recovery
The recovery of ethanol is achieved through a series of distillation processes, which can recover up to 95% of the ethanol. To achieve absolute alcohol (100%), an azeotropic mixture with 5% water must be created. Subsequently, 5% of the water within the azeotropic combination of ethanol, water, and benzene is removed. The final steps involve the elimination of benzene to obtain pure ethanol.
Microorganisms Used in Ethanol Production
Various microorganisms are employed in alcohol production, including yeasts (e.g., Saccharomyces cerevisiae, S. uvarum, S. carlsbergensis, Candida brassicae, C. utilis, Kluyveromyces fragilis, K. lactis) and bacteria (Zymomonas mobilis). Commercial ethanol production primarily relies on Saccharomyces cerevisiae, although other microorganisms have been explored for specific applications.
Recent experimentation with Schizosaccharomyces has shown promising outcomes, especially when dealing with milk whey as a feedstock. Additionally, certain yeast strains, along with Fusarium and Bacillus, have demonstrated the ability to convert pentose carbohydrates into ethanol.
Impact of Ethanol Concentration
It’s worth noting that high concentrations of ethanol can hinder yeast growth, affecting ethanol biosynthesis. This dynamic highlight the importance of carefully managing ethanol concentration during the fermentation process.
Alternative Bacterium in Ethanol production: Zymomonas mobilis
Zymomonas mobilis, a bacterium, offers advantages over yeast due to its superior sugar concentration osmotic tolerance, specialized growth rate, and relatively high resistance to ethanol.
References
- Desbandhu College PDF
- Microbiology Note – Alcohol (EtOH) Production
- UOMustansiriyah PDF
- Microbiology Notes – Ethanol Production, Recovery, and Uses