Tetracycline is an important medicine that was found by accident in the 1940s. It’s like a superhero antibiotic that fights many types of bacterial infections, like ones you can get from having sex, bladder problems, or breathing issues. Tetracycline works by stopping bacteria from making proteins, and it can fight both weak and strong bacteria. In this article, we’ll learn more about tetracycline production, including its history, what it’s made of, how it’s used in medicine, and how it helps us fight diseases caused by bacteria. We’ll also explore how it’s made and how it works inside our bodies, showing why it’s still important today.
Introduction of tetracycline
A crucial role in the development of medicine has been played by the broad-spectrum antibiotic tetracycline. Its discovery in the 1940s completely changed how bacteria were treated for a variety of bacterial diseases, including sexually transmitted infections, urinary tract infections, and lung infections.
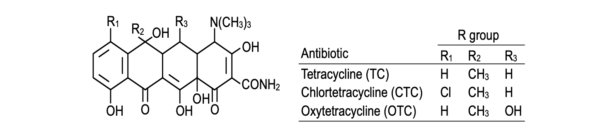
Tetracycline is a member of a class of antibiotics that prevents the synthesis of bacterial proteins, making it effective against both Gram-positive and Gram-negative bacteria.
Tetracycline has good oral absorption and low toxicity. It needs a large dosage and is bacteriostatic. Treatment for shigellosis, salmonellosis, typhoid fever, brucellosis, etc. involves the administration of this antibiotic.
Additionally, it is utilized in feed to get rid of parasites (and aid animals in gaining weight). The antibiotic is also used to preserve fish (tetracycline is used to disinfect the ice in which the fish is kept).
Tetracycline has a lengthy history in medicine, but few people are using it now because of the rise of antibiotic resistance and the accessibility of newer antibiotics. Nevertheless, it continues to be a focus of research for its potential applications outside of conventional antibiotic therapy and is an essential tool in several medical situations.
Learn more about
Mechanism of Action and Antimicrobial Activity of Macrolides (thesciencenotes.com)
Some’s members of tetracycline

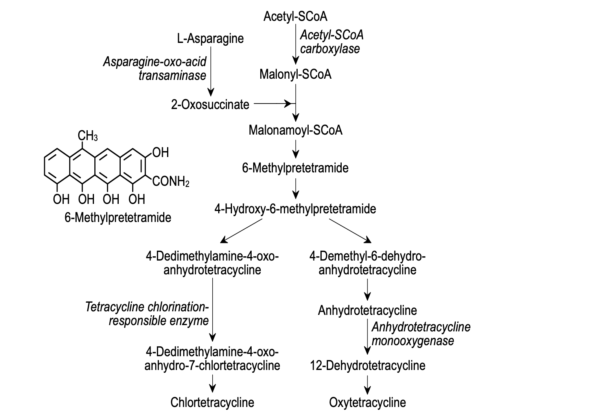
Biosynthesis of tetracycline
Biosynthesis of tetracyclines, particularly in the latter stages, has been studied with the use of mutant cultures. The involvement of pretetronid intermediates in the synthesis has been established. Tetracyclines are created from these intermediates by the bacterium Streptomyces aureofaciens.
Learn more about
Production of tetracycline
Both chemical and microbiologic production methods can be used to make tetracycline. All tetracyclines are produced by the same types of microbes. However, compared to other tetracyclines, chlortetracycline synthesis is much simpler. Tetracycline production is particularly sensitive to the amount of chloride in the medium because it triggers the formation of chlortetracycline rather than tetracycline.
Tetracycline antibiotics are produced by a complex fermentation process involving microbes, usually strains of the bacterium Streptomyces. Here’s a simplified overview of the production process:
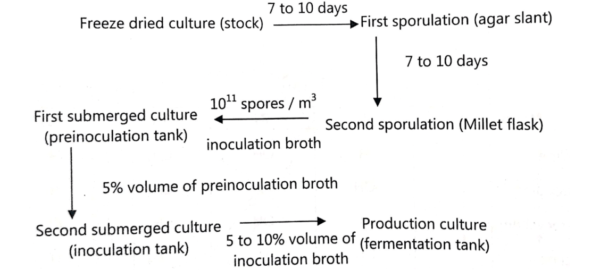
Microorganism Selection:
For cultivation, Streptomyces bacterial strains that can produce tetracycline compounds are isolated and selected.
Inoculation:
In a laboratory, a selected strain of Streptomyces is cultured on an agar medium that is rich in nutrients. The inoculum for the fermentation process is used once a pure culture has been obtained.
During inoculum build-up, the organism remains in the shake-flask for 24 hrs at 28°C. The most effective propagator makes use of the same kind of medium. Propagation is carried out for 19–24 hours at a pH of 5.2–6.2 with an addition of about 5% inoculum.
From the final propagator, 2-10% of the inoculum is transferred to the main fermenter. The nominal capacity of the fermenter is between 25,000 and 75,000 liters. Sterilized medium is used for fermentation (121°C for 1-2 hours).
Fermentation:
The Streptomyces culture is grown in large-scale fermentation tanks, commonly referred to as bioreactors. The temperature, pH, and aeration are all controlled in these tanks. The bacteria are raised in a nutritional broth that also includes additional essential nutrients like carbohydrates and amino acids.
At 28°C, the primary fermentation lasts 60–65 hours. The pH is between 5.8 and 6. It is a fermentation that is submerged that needs aeration at a rate of 0.5-2 vol/vol/min.
Mechanical agitators are used for agitation. As an antifoam, lard is employed. Since glucose inhibits catabolism, it is typically not employed in the primary fermentation. Around 15000 units per milliliter of output.
Tetracycline production:
Tetracycline compounds are produced by the Streptomyces bacterium as secondary metabolites during the fermentation process. Depending on the strain and conditions, the precise chemical composition of the tetracycline generated can change.
Harvesting:
The fermentation broth is removed out of the process once enough tetracycline has been produced. Usually, the broth also contains tetracycline in addition to additional components.
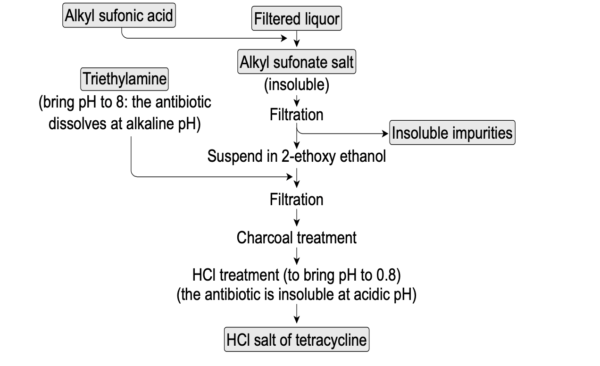
Purification:
To separate the tetracycline component from contaminants and other byproducts, the recovered broth passes through a number of purification processes. These procedures frequently involve chromatography, precipitation, and filtering.
Mode of action of tetracycline
Numerous enzymatic reactions necessary for the survival of bacterial cells are inhibited by tetracyclines. The synthesis of proteins is the biological process that is hindered most delicately.
Tetracyclines prevent the aminoacyl tRNA from binding to the RNA-ribosome complex by specifically binding to the bacteria’s 30S ribosome.
It simultaneously inhibits other steps of protein biosynthesis Tetracycline can also change the cytoplasmic membrane, which leads to the leakage of nucleotides and other substances from the cell. Instead of destroying the germs directly, this inhibits them.
References
- Essentials of Industrial Microbiology (PDF) https://foodtechnotes.com
- Tetracycline Production: A Comprehensive Guide (https://www.scielo.br)
- Production of Tetracycline: A Detailed Overview (https://biologyease.com)
- “Advances in Tetracycline Production” – Published in Biotechnology and Bioengineering (https://onlinelibrary.wiley.com/doi/abs/10.1002/bit.260330811)
- Fermentation Production of Tetracycline: A Patent Perspective (https://www.hovione.com)