Triple sugar iron agar (TSI) / TSIA Test is a highly versatile and indispensable tool in the field of microbiology. With its ability to differentiate enteric bacteria based on carbohydrate fermentation and hydrogen sulfide production, TSI plays a crucial role in bacterial identification, clinical diagnostics, and research endeavors. This comprehensive medium contains lactose, sucrose, glucose, ferrous sulfate, and phenol red, providing a rich substrate for assessing bacterial metabolic activities.
Principle of TSIA Test
The principle underlying the Triple Sugar Iron (TSI) agar test revolves around its differential capabilities to discern between various Gram-negative enteric pathogens based on their metabolic activities. This medium contains three crucial components: triple sugars (lactose, sucrose, and glucose), a pH indicator (phenol red), and ferrous sulfate. Each component serves a specific purpose in facilitating the differentiation of bacterial strains.
Triple Sugars (Lactose, Sucrose, and Glucose):
These carbohydrates serve as substrates for bacterial fermentation. Bacteria with the enzymatic machinery to metabolize these sugars will utilize them as energy sources, leading to the production of acidic byproducts. The utilization of different sugars produces characteristic color changes in the medium, aiding in the identification of metabolic capabilities.
pH Indicator (Phenol Red):
Phenol red acts as a pH indicator, changing color in response to alterations in the pH of the medium. Initially, the TSI agar appears reddish-orange, indicating a neutral pH. As bacteria ferment sugars and produce acidic byproducts, the pH of the medium decreases, causing phenol red to shift from yellow (acidic pH) to red (basic pH). This color change provides visual cues about the fermentation patterns of the bacteria.
Ferrous Sulfate:
Ferrous sulfate serves a dual purpose in the TSI agar test. Firstly, it acts as a substrate for the detection of hydrogen sulfide (H2S) production by certain bacterial species. Bacteria capable of reducing sulfur compounds present in the medium produce hydrogen sulfide gas, which reacts with ferrous sulfate to form a black precipitate of ferrous sulfide. This blackening of the medium serves as an indicator of H2S production. Secondly, ferrous sulfate also aids in detecting gas production by bacteria. As gas is produced during sugar fermentation, it may manifest as bubbles or fissures in the medium, providing additional diagnostic information.
By incorporating these components into the TSI agar medium, microbiologists can exploit the metabolic diversity of enteric pathogens to differentiate between various bacterial strains. The interpretation of TSI agar results relies on observing color changes, gas production, and the formation of black precipitates, enabling microbiologists to make informed decisions regarding bacterial identification and classification.
Interpreting TSIA Test Results:
The interpretation of TSIA test results involves careful observation of color changes and precipitate formations within the medium. These observations provide valuable insights into bacterial metabolic profiles. The table below outlines the interpretation of different TSI result combinations:
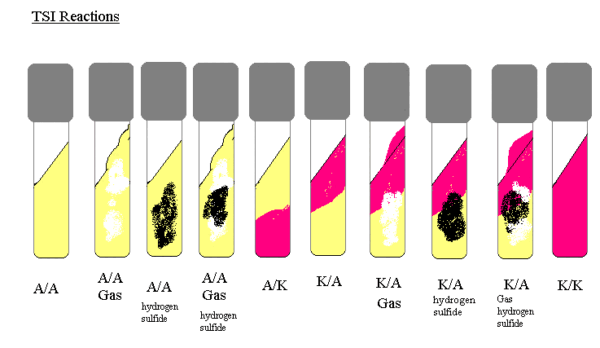
| Result (slant/butt) | Symbol | Interpretation |
| Red/Yellow | K/A | Glucose fermentation only, peptone catabolized. |
| Yellow/Yellow | A/A | Glucose and lactose and/or sucrose fermentation. |
| Red/Red | K/K | No fermentation, peptone catabolized. |
| Yellow/Yellow with bubbles | A/A,G | Glucose and lactose and/or sucrose fermentation, Gas produced. |
| Red/Yellow with bubbles | K/A,G | Glucose fermentation only, Gas produced. |
| Red/Yellow with bubbles and black precipitate | K/A,G,H2S | Glucose fermentation only, Gas produced, H2S produced. |
| Yellow/Yellow with bubbles and black precipitate | A/A,G,H2S | Glucose and lactose and/or sucrose fermentation, Gas produced, H2S produced. |
| Red/Yellow with black precipitate | K/A,H2S | Glucose fermentation only, H2S produced. |
| Yellow/Yellow with black precipitate | A/A,H2S | Glucose and lactose and/or sucrose fermentation, H2S produced. |
Expected Results and Examples of Bacteria in TSIA test:
- Red Slant/Yellow Butt (K/A):
- Example Bacteria: Citrobacter freundii, Citrobacter koseri, Morganella morganii.
- Yellow Slant/Yellow Butt (A/A):
- Example Bacteria: Escherichia coli, Enterobacter aerogenes, Klebsiella oxytoca.
- Red Slant/Red Butt (K/K):
- Example Bacteria: Acinetobacter spp., Pseudomonas spp.
- Yellow Slant/Red Butt with Gas (A/A, G):
- Example Bacteria: Escherichia coli, Enterobacter aerogenes, Klebsiella pneumoniae.
- Red Slant/Yellow Butt with Gas and Black Precipitate (K/A, G, H2S):
- Example Bacteria: Citrobacter freundii, Proteus mirabilis.
- Yellow Slant/Yellow Butt with Gas and Black Precipitate (A/A, G, H2S):
- Example Bacteria: Proteus mirabilis, Proteus vulgaris.
- Red Slant/Yellow Butt with Black Precipitate (K/A, H2S):
- Example Bacteria: Citrobacter freundii, Salmonella spp.
- Yellow Slant/Yellow Butt with Black Precipitate (A/A, H2S):
- Example Bacteria: Proteus mirabilis, Proteus vulgaris.
Distinctive Features of TSI Results:
- Color Changes: Indicate the utilization of specific sugars and pH alterations.
- Gas Production: Manifests as bubbles or cracks in the agar medium.
- H2S Production: Leads to a black precipitate formation in the medium.
Practical Considerations:
- Incubate TSI tubes at 37°C for 18-24 hours for optimal results.
- Prolonged incubation may distort interpretations, affecting result reliability.
TSIA Recipe:
Ingredients:
- Pancreatic digest of casein USP: 10.0 g
- Peptic digest of animal tissue USP: 10.0 g
- Glucose: 1.0 g
- Lactose: 10.0 g
- Sucrose: 10.0 g
- Ferrous sulfate or ferrous ammonium sulfate: 0.2 g
- Sodium chloride (NaCl): 5.0 g
- Sodium thiosulfate: 0.3 g
- Phenol red: 0.024 g
- Agar: 13.0 g
- Distilled water: 1,000 mL
Instructions:
- Mix all ingredients in distilled water.
- Adjust pH to 7.3.
- Boil until dissolved.
- Sterilize by autoclaving at 121°C for 15 minutes.
- Cool and dispense into tubes with slanted position.
- Solidify for use in bacterial culture.
Storage: Keep in cool, dry place away from sunlight.
This recipe creates TSIA agar, facilitating detection of carbohydrate fermentation and H2S production in bacteria, aiding in microbial identification.
Significance of TSIA test in Microbiological Diagnostics and Research:
The Triple Sugar Iron agar test holds immense significance in microbiological diagnostics and research for several reasons:
- Bacterial Identification: TSI enables the differentiation of bacterial strains based on their metabolic capabilities, aiding in bacterial identification and classification.
- Clinical Relevance: TSI results inform clinical interventions, particularly in diagnosing gastrointestinal infections caused by enteric bacteria, guiding treatment strategies and preventing disease spread.
- Microbial Ecology Studies: TSI agar test is instrumental in microbial ecology studies, providing insights into the diversity and metabolic activities of bacterial communities in different environments.
- Research Tool: TSI agar test serves as a valuable research tool for studying bacterial metabolism, adaptation mechanisms, and virulence factors, contributing to our understanding of bacterial physiology.
- Quality Control: TSI agar test is used for quality control purposes in clinical and industrial settings, ensuring the purity and safety of various products by detecting potential enteric pathogens.
Conclusion:
In summary, the Triple Sugar Iron agar test stands as a cornerstone technique in microbiology, offering valuable insights into bacterial metabolic profiles and aiding in bacterial identification, clinical diagnostics, and research endeavors. Through meticulous interpretation and adherence to best practices, TSI agar test continues to play a pivotal role in advancing our understanding of microbial communities and their implications for human health.
Learn More;