- Chromatography is derived from the Greek word ‘chroma’ means ‘color’ and ‘graphein’ means writing or recording.
- In 1890, Mikhail Tsvet, a Russian Italian Botanist invented an earliest form of true chromatography technique for the separation of plant pigmentation.
- But later, evolution of paper chromatography stroked and improved by Raphael E Liesegang in 1927. Archer Martin and Richard Synge again popularized it and further developed gas chromatography in collaboration with Anthony James.
- Chromatography separates a component of mixture which is dissolved in a substance called the mobile phase and is carried out by a second substance called the stationary phase.
- It separates a chemical mixture into an individual component and helps in analysis of the particular compound.
Chromatography is generally carried out by organic chemist and biochemists for analysis, isolation and purification.
Working principle of Chromatography
- Chromatography is a method of physical separation in which components of mixture gets separated on two phases.
- One of the phase is the immobile porous bed bulk liquid which is called stationary phase and the other phase is the mobile fluid that flows over the stationary phase under gravity.
- During the movement of the sample, a separated result is formed by the repeated desorption and sorption in the direction of the mobile phase migration.
- Several key factors are responsible on the separation process like partition between liquid-liquid, affinity between molecular weight and characteristics related to liquid-solid adsorption.
- An interaction between the molecules are physical and involves weak chemical bonds like dipole-dipole interaction and hydrogen bond formation and adhere to the stationary components.
- Components that adhere strongly to the stationary phase moves slowly than those who adhere weakly.
Types of Chromatography
Commonly used chromatographic technique differs in the kind of stationary and mobile phase they use:
- Paper chromatography
- Thin layer chromatography
- Liquid column chromatography
- Size exclusion chromatography
- Ion exchange chromatography
- Affinity chromatography
- Gas chromatography
- High performance liquid chromatography
Paper chromatography
In a paper chromatography, separation of the mixture is performed on a paper strip which is a stationary phase and a liquid solvent acts as a mobile phase.
How does Paper Chromatography work?
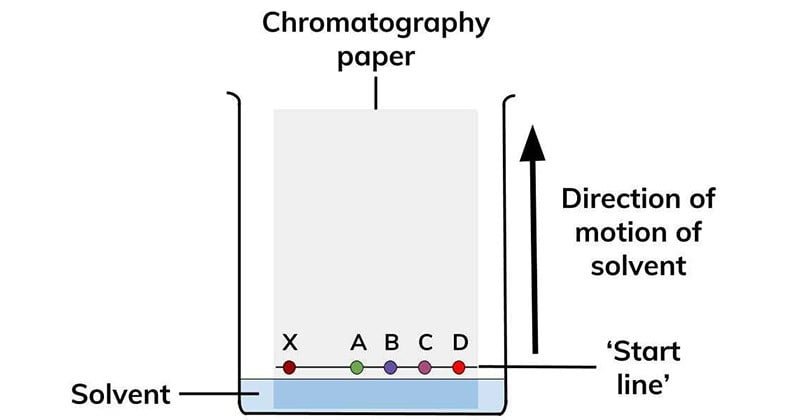
- A drop of mixture is placed on one end of the paper and dried. Then the paper is dipped into the solvent up to the spot.
- In the paper chromatography, component separates in two ways:
In paper adsorption chromatography, stationary phase and mobile phase molecules act based on the degree of interaction. Higher affinity molecules are adsorbed for a long time where movement of speed is decreased.
However, low affinity molecules move faster thus, molecules are separated.
In paper partition chromatography, the moisture on the cellulose layers acts as a stationary phase where the mobile phase molecules are strongly absorbed hence, the molecules gets separated.
- During the separation of molecules, retention factor is determined as;
Retention factor (Rf) = Distance travelled by solute
Distance travelled by solvent
Thin layer chromatography (TLC)
Thin layer chromatography works on the basis of solid-liquid partition where silica or alumina plate used is a stationary phase and liquid as a mobile phase.
Basically, TLC is a technique that is used to separate non- volatile mixtures.
How does Thin Layer chromatography work?
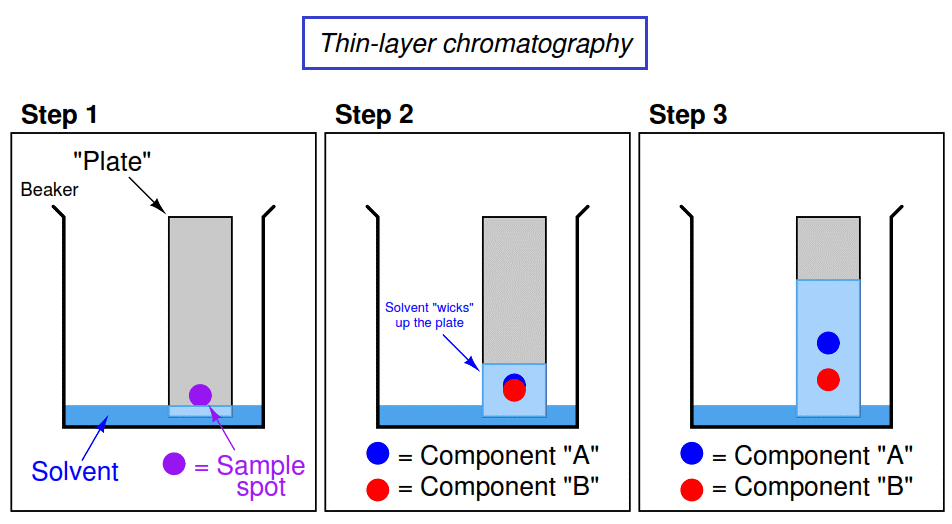
A thin layer of silica gel or cellulose is used as an adsorbent and the solvent mixture as a mobile phase. The mixture of the solution is placed at a distance of 2cm above one end of the plate. Then the plate is placed in a jar containing a fluid solvent.
Moving phase is drawn up by the capillary action where the mixture rises up the plate carrying different components.
The plate is taken out after the mobile solvent reaches the top and its retention factor is calculated.
Liquid column chromatography
Liquid column chromatography is a technique used to separate when a molecules or ions are dissolved in a solvent, it separate the components using a column of adsorbent packed glass tube.
How does Liquid column chromatography work?
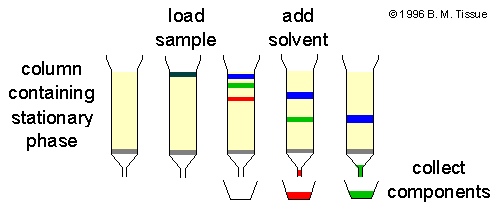
- The slurry or powdered silica acts as a stationary phase which is loaded on a column or a paper and the sample is added to the liquid in the chromatographic system which acts as a mobile phase.
- The degree of the adsorption depends upon the affinity of the molecules. If the components have a high affinity, the molecule moves down the column faster but low degree affinity molecules move slowly and remains at the top.
- High performance liquid chromatography (HPLC) is a modified version of the liquid column chromatography.
Size exclusion chromatography
From its given name, size exclusion chromatography is a method where the molecules in a solution are separated in terms of their size or weight.
It is also known as gel filtration, gel permeation and molecular sieve chromatography.
How does size exclusion chromatography work?
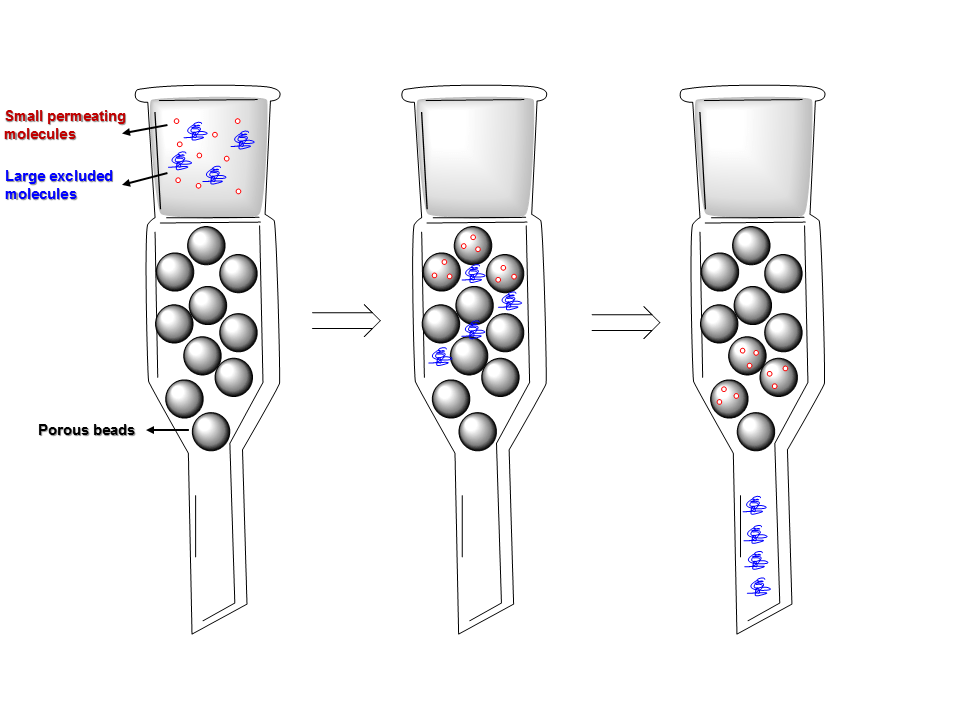
- The semi permeable porous polymer gel bead is a stationary phase molecules that is filled in a column and the sample is mixed with mobile phase liquid.
- The molecules are partitioned on the basis of their respective sizes. Molecules with small size moves fast out of the column while the large size molecules stay at the top.
- If the aqueous solution is used in mobile phase then it is gel filtration. Similarly, if an organic solvent is used, it is gel permeation chromatography.
Ion exchange chromatography
In ion exchange chromatography, the separation is between the charged molecules where solid polymeric ion exchange resins are used.
This technique is mostly used to purify the drinking water.
How does Ion exchange chromatography work?
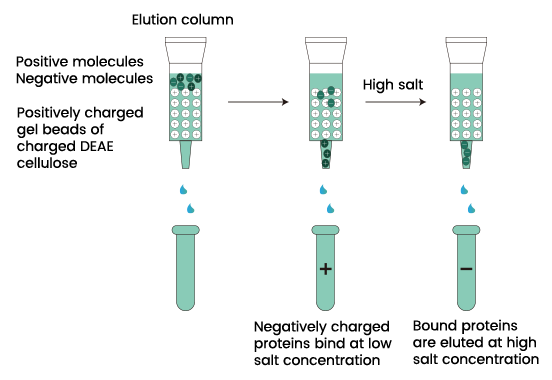
- In a column, charged ion resins are packed which is taken as stationary phase. The mixture with the charged particles when passed through the column binds to the oppositely charged resins.
- If a negative charged molecule is used, it binds to the positively charged resins and vice versa.
- Then an appropriate buffer is used to separate the complex charged molecules and resins.
Affinity chromatography
The mixture is separated on the basis of affinity between the absorbent and the ligand.
Absorbent is the separation material where the ligand is the desired components in a mixture
How does Affinity Chromatography work?
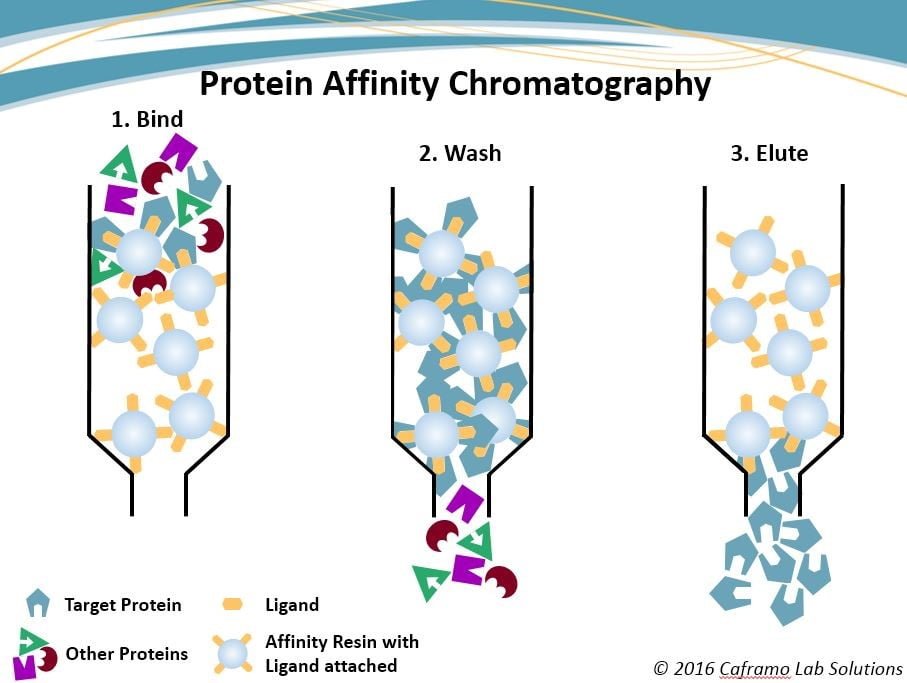
- Agarose or cellulose is loaded in a column as a solid support. The mixture of mobile phase is placed at a constant rate.
- The ligand molecule complex is formed during the process and rests of the components are eluted out with the mobile phase.
- Components with the stationary phase are obtained by changing pH and its ionic strength.
Gas chromatography
Gas chromatography is a technique of separation of chemical components which are usually organic molecules or gases and determines their presence.
How does Gas Chromatography work?
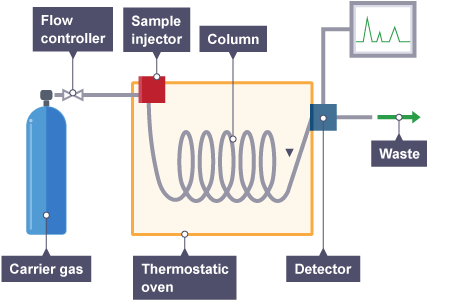
- It is different from other chromatography because the gas acts as a mobile phase and the separation is vapor.
- The sample is injected into the column which is either gas or liquid.
- Helium is usually used as a carrier gas as a mobile phase which moves in the analyte through the column at their own rate.
- The components are collected and their retention time is determined.
High Performance Liquid chromatography
HPLC is a modified version of liquid column chromatography. As smaller particles size <10 microns are separated with a high pressure.
How does HPLC work?

- Cellulose or silica is taken in a column of glass tube where sample mixture of mobile phase is introduced from the top of the column and a high pressure is passed through a pump at a constant rate.
- The components are separated on the basis of different adsorption.
- High affinity molecules remain adsorbed for a long time and moves slowly while low affinity molecules move faster.
- The process is similar as of liquid column chromatography but only a solvent is forced under high pressure up to 400atm.
Application of chromatography
- In molecular biology, protein separation, insulin and enzyme purification and some biochemical processes, HPLC is used. It is also applied in the study of proteomics and metabolomics.
- In chemical industries, HPLC and GC is used for the water and air purification and in pesticides and oil testing.
- In pharmaceutical industries, chemicals of different weight and sizes are separated and identified and also detects the unknown compounds.
- In food industries, toxins and food additives are determined.
References:
- https://www.khanacademy.org/science/class-11-chemistry-india/xfbb6cb8fc2bd00c8:in-in-organic-chemistry-some-basic-principles-and-techniques/xfbb6cb8fc2bd00c8:in-in-methods-of-purification-of-organic-compounds/a/principles-of-chromatography
- https://microbenotes.com/chromatography-principle-types-and-applications/
- https://microbenotes.com/types-of-chromatography/
- Poole, C. F. (2000). CHROMATOGRAPHY. Encyclopedia of Separation Science, 40–64.