Stable cell lines play a crucial role in advancing modern scientific research. Unlike transient transfection—where gene expression is temporary and lost during cell division—stable cell lines enable prolonged, consistent, and reproducible gene expression. This capability is vital for drug discovery, functional genomics, and the manufacturing of biopharmaceuticals.
This comprehensive guide explores the significance of stable cell lines, the molecular mechanisms behind their creation, and a detailed 10-step protocol for establishing lines that constitutively express GFP-tagged proteins.
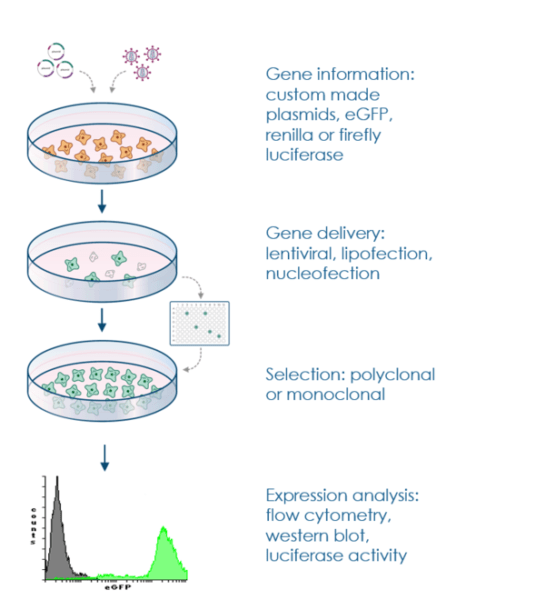
1. Exploring the Significance of Stable Cell Lines
In the landscape of cell biology, stability is the gold standard for long-term experiments. Stable cell lines serve as robust platforms facilitating the exploration of protein expression, localization, and functional dynamics within a controlled cellular context.
These specialized lines are crafted by introducing a Gene of Interest (GOI) into host cells and selectively nurturing those that have seamlessly integrated that gene into their genetic blueprint. The primary objective is to cultivate a cellular population where a substantial proportion consistently showcases the fusion protein’s expression. This concerted effort allows researchers to delve deep into intricate behaviors, such as protein-protein interactions, signaling cascades, and organelle trafficking, without the “noise” associated with fluctuating expression levels found in transient systems.
2. Molecular Methods for Generation: Episomal vs. Genomic Integration
There are two primary architectural approaches to maintaining a transgene within a cell line:
A. Episomal Maintenance
This involves introducing a GOI using specialized vectors designed to be retained as episomes (extrachromosomal DNA) within the nucleus. These vectors often contain viral elements (like those from EBV or SV40) that allow them to replicate alongside the host’s genome.
Pros: High copy numbers and rapid generation.
Cons: Potential for loss over many passages and limited suitability for certain long-term clinical applications.
B. Direct Genomic Integration
This is the standard for “true” stable cell lines. The DNA is integrated directly into the host cell’s genome, often via non-homologous end joining (NHEJ) or targeted recombination.
Pros: Maximum stability, inherited by all daughter cells, and highly preferred for longitudinal studies.
Cons: Lower initial efficiency and the risk of “positional effects,” where the integration site influences the expression level of the transgene.
3. The Critical Role of Selection Markers
The successful creation of stable cell lines hinges on the use of a selection marker. Because the probability of a cell spontaneously integrating foreign DNA into its genome is extremely low (often 1 in $10^4$ or $10^5$ cells), researchers must use a “survival of the fittest” strategy.
Selection markers are typically resistance genes to antibiotics. When the selective agent is added to the culture medium, only the cells that have integrated the plasmid—and therefore the resistance gene—will survive.
| Marker | Antibiotic Agent | Mechanism |
| NeoR / KanR | G418 (Geneticin) | Blocks polypeptide synthesis by inhibiting elongation. |
| PuroR | Puromycin | Causes premature chain termination during translation. |
| HygR | Hygromycin B | Inhibits protein synthesis by interfering with translocation. |
| DHFR | Methotrexate | Used in DHFR-deficient cells for gene amplification. |
| GS | Methionine Sulfoximine | Glutamine Synthetase system for high-yield protein production. |
To ensure success, the marker gene must be co-expressed with the target protein. This is usually achieved by placing both genes on the same plasmid (often separated by an IRES or 2A peptide sequence) or by co-transfecting two separate vectors at a specific ratio.
4. Pre-Experimental Preparation: Culture Conditions
The physiological state of the host cell type is pivotal. Even the most sophisticated plasmid will fail if the host cells are stressed or over-passaged.
Supplier Compliance: Always follow recommendations from repositories like ATCC or ECACC.
The “Log Phase” Rule: Cells should be in the logarithmic growth phase at the time of transfection. This usually requires passaging cells two days before the experiment.
Passage Limits: To maintain genetic integrity and transfection receptivity, it is generally recommended to keep passage numbers below 30. Older cells may develop “transfection resistance” or undergo phenotypic drift.
5. The 10-Step Stable Cell Line Generation Procedure
Establishing a stable line is a marathon, not a sprint. The process typically takes 4 to 12 weeks depending on the growth rate of the cell line.
Step 1: Select the Host Cell Line
Choose a cell line relevant to your research. Common models include:
HEK293: Excellent transfection efficiency; great for general protein work.
CHO (Chinese Hamster Ovary): The industry standard for biopharmaceutical production.
HeLa: Robust and widely used in cancer research.
Jurkat: Ideal for immunology and T-cell signaling.
Step 2: Plasmid Construction
Design a vector that contains your GOI (e.g., a GFP-tagged protein) under a strong promoter (like CMV or EF1$\alpha$) and a selectable marker. Consider linearizing the plasmid with a single-cut restriction enzyme in a non-essential region; this can significantly increase the rate of stable integration.
Step 3: Transfection
Introduce the DNA into the cells. The choice of method depends on the cell type:
Lipofection: Best for common adherent cells.
Electroporation: Necessary for hard-to-transfect primary cells or suspension cells.
Viral Transduction (Lentivirus/Retrovirus): Often provides the highest stability and is excellent for non-dividing cells.
Step 4: Antibiotic Selection
48 hours post-transfection, replace the media with fresh media containing the selection agent. The concentration should be determined beforehand via a Kill Curve. Over the next 7–14 days, non-transfected cells will die off, leaving behind small clusters of resistant cells.
Step 5: Single-Cell Cloning
A “mixed population” (pool) is often genetically undefined. To get a truly stable and homogenous line, you must perform limiting dilution. Cells are plated in 96-well plates at a density of ~0.5 cells per well. This statistical approach ensures that any resulting colony grew from one single cell.
Step 6: Expansion and Screening
Once colonies reach sufficient confluency, expand them into larger vessels (24-well plates, then T-25 flasks). Screen the clones for the GOI using:
Fluorescence Microscopy: For GFP-tagged proteins.
Western Blotting: To confirm protein size and expression levels.
RT-qPCR: To quantify mRNA levels.
Step 7: Verification and Characterization
Confirm that the integration hasn’t altered the cell’s biology. Perform functional assays, check growth kinetics, and ensure that the GFP tag does not interfere with the protein’s native biological activity or localization.
Step 8: Cryopreservation
Once a high-expressing, stable clone is identified, freeze it immediately. Create a “Master Cell Bank” (MCB) and a “Working Cell Bank” (WCB) using early-passage cells to protect against future contamination or genetic drift.
Step 9: Detailed Documentation
Maintain meticulous records of plasmid maps, transfection parameters, the specific selection agent batch, and cloning lineage. This is essential for reproducibility and potential patent applications.
Step 10: Routine Maintenance
Even “stable” lines can lose expression over time due to epigenetic silencing (e.g., DNA methylation). Regularly maintain selective pressure by adding a low dose of antibiotic to the growth media and monitor expression levels periodically via flow cytometry.
6. Key Technical Considerations for Optimization
To achieve a 70% or higher success rate in stable line generation, observe these three technical nuances:
The Kill Curve: Never guess the antibiotic dose. Test a range of concentrations on wild-type cells. The optimal dose is the lowest concentration that kills 100% of cells within 7 days.
Reporter Plasmids: Before the “real” experiment, optimize your transfection settings using a simple GFP reporter plasmid to ensure you are achieving at least 30-50% transient efficiency.
Linearization Strategy: Circular plasmids can integrate, but they often break in the middle of your GOI. Linearizing the plasmid at a site far from your promoter and GOI ensures the “business end” of your DNA remains intact during recombination.
7. Diverse Applications of Stable Cell Lines
The ability to produce proteins consistently has revolutionized several fields:
Biopharmaceutical Production: Virtually all therapeutic monoclonal antibodies are produced in stable CHO or NS0 cell lines.
Drug Screening: High-throughput screening (HTS) uses stable lines expressing disease-related targets to test thousands of chemical compounds.
Cancer Research: Stable lines mimicking oncogenic mutations help scientists understand how tumors develop and resist treatment.
Neurobiology: Expressing proteins like tau or alpha-synuclein in stable neuronal models is critical for Alzheimer’s and Parkinson’s research.
Viral Studies: Lines expressing specific receptors (like ACE2 for SARS-CoV-2) are indispensable for studying viral entry and vaccine efficacy.
8. Frequently Asked Questions (FAQs)
Q: How long does a “stable” cell line remain stable?
A: If correctly generated and maintained under selective pressure, they can remain stable for dozens of passages. However, it is best practice to restart from a frozen WCB every 2-3 months to avoid “phenotypic drift.”
Q: Why do my cells look green at first but lose color after two weeks?
A: This is a common issue. You are likely seeing transient expression from plasmids that entered the cell but did not integrate into the genome. Once the cells divide, the non-integrated plasmids are diluted out. Only selection will rescue the few cells with genomic integration.
Q: Can I use two different antibiotics for two different genes?
A: Yes, this is called dual selection. You can establish a line expressing two different proteins by using, for example, Neomycin resistance for the first and Puromycin for the second.
9. Conclusion
Establishing a stable cell line is a foundational skill in cell biology that bridges the gap between basic research and industrial application. By following a rigorous 10-step protocol—from careful cell selection and plasmid linearization to meticulous single-cell cloning—researchers can create high-quality, reproducible models. As CRISPR and other gene-editing technologies evolve, the precision of stable cell line generation will only continue to improve, further uncovering the intricacies of cellular life.
References
Harvard Medical School – Fly Cell Lines.
Addgene – Protocol for Generating Stable Cell Lines.
NCBI – Guidelines for Genomic Integration and Stable Expression.
Mirus Bio – Technical Guide: Transitioning from Transient to Stable Transfection.
ATCC – Best Practices for Maintaining Cell Line Identity and Viability.