Plant cells have potential to grow as a plant. This potential of the plant is called totipotency. Any component of the plant can grow into a complete plant because of this ability. It is possible to artificially develop plant tissue or cells in a lab setting. Plant tissue culture is a method for cultivating plant cells, tissues, or organs under aseptic conditions in a nutritional medium that has been artificially created.
Haberlandt (1896) was the first person to culture the isolated vegetative cells of higher plants in simple nutrient solutions. Although Haberlandt was able to maintain the cells in the nutrient media, the cell division was not recorded until much later. Murashighe (1961) demonstrated the technique of in vitro culture of propagation of various plant species. Guha and Maheshwari (1964) first of all successfully reported anther culture of Datura.
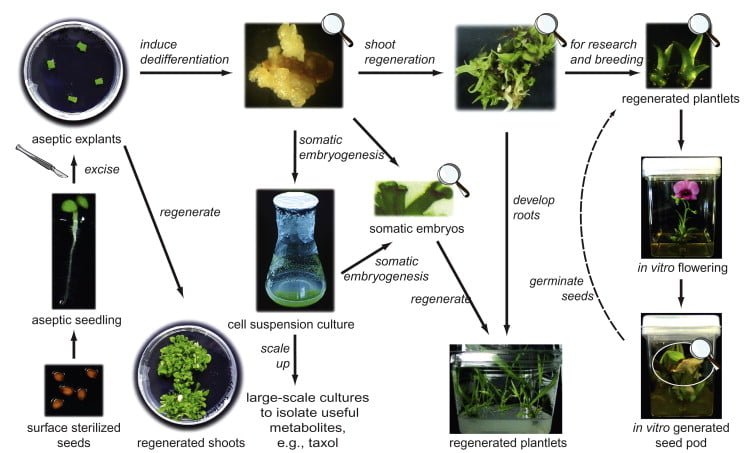
Plant tissue culture for biotechnology,
Types of plant tissue culture
Due to totipotency, every single component of a plant has the capacity to develop into a plant. For tissue culture, plant parts like roots, buds, shoots, leaves, anthers, ovules, etc. can be utilized as explants. On the basis of the explants used, plant tissue culture can be of the following steps:
Shoot culture
This kind allows for the use of a variety of adventitious, apical, and axillary shoots as an explant for plant tissue culture. To directly grow plants, these shoots can be cultivated on a plant tissue culture medium. This process is referred to as clonal propagation.
Shoots → Plants
Callus culture
In this process, a large number of plantlets can be obtained from the callus. The callus is a mass of undifferentiated, dispersed cells. When poured on solidified culture medium, it forms around the cut edges of the plant segments (explants) of the plant organs. This approach allows for the rapid production of a large number of plantlets.
Tissue → Callus → Plants
Embryo culture
In this culture, ovules are excised and embryos are dissected out and transferred to culture medium. Knudson (1992) successfully grew orchid embryos into plantlets by culturing them on agar medium containing sugar.
Embryo → Plants
Meristem-tip culture
It is the technique to culture meristem aseptically in an artificial condition in laboratory medium. Meristem is the apical growing part of plants which always keeps on multiplying. It is also a disease-free region. Meristem-tip culture produces virus free plants. Therefore, this technique has its major application in eliminating various diseases.
Anther culture
Anther culture is a means of obtaining haploid plants from the pollen grains. In this method, anthers are used as explants for the plant tissue culture. This process is called androgenesis. Guha and Maheshwari (1964) first of all, successfully reported the anther culture of Datura. Anther culture is of special interest to geneticists and plant breeders as it produces haploid or homozygous plants.
Anther → Callus → Plants
Protoplast culture
Protoplast is a cell without a cell wall (naked plant cell). The cell wall is dissolved by enzymes called cellulase and pectinase. Protoplasts can be isolated from leaves, stem callus and from pollen grains. These can be grown on the agar containing nutrients. A complete plant can be obtained from a single protoplast. Protoplast is useful in obtaining germ-free plants, introducing foreign genes, and fusion of two incompatible plants.
PLANT TISSUE CULTURE MEDIA COMPONENTS
In spite of the fact that there are a variety of ingredients and additives that can be used in plant micropropagation media, the majority of them fall into one of eight categories:
Water- To ensure that no minerals or other pollutants have an impact on plant growth, ultrapure water is needed. Reverse osmosis or distillation are both used to achieve this.
Nutrient salts (micro and macronutrients): Macronutrients, such as nitrogen, phosphorus, potassium, calcium, magnesium, and sulfur, are needed in higher concentrations (1–60 mM). Micronutrients, such as iron, manganese, zinc, boron, copper, molybdenum, cobalt, and iodine, are needed in lesser amounts (0.1–100 M).
Vitamins: Thiamine, nicotinic acid, pyridoxine, and myo-inositol to stimulate certain metabolic functions.
Amino acids: As a conveniently available supply of nitrogen, different amounts of single amino acids like glycine, glutamine, or adenine as well as amino acid combinations like casein hydrolysate may be added to the media.
Carbohydrates: Sucrose is the most widely utilized carbohydrate source, while others include glucose, fructose, and maltose. Unlike in the natural environment, most in vitro plants are unable to synthesize their own carbohydrates from light, water, and carbon dioxide.
Gelling agents: The goal of gelling agents is to give the plant being cultivated physical support. Agar is frequently used.
Growth regulators (hormones): Auxins, cytokinins, gibberellins, and abscisic acid are used. While cytokinins favor axillary and adventitious shoot production and growth, auxins tend to promote root and callus initiation and growth. Abscisic acid can boost and improve the general quality of somatic embryogenesis, and gibberellic acid is utilized to encourage shoot elongation.
Organic supplements
Application of Plant tissue culture in Agriculture
- Micropropagation: It is the rapid vegetative multiplication of plant materials for agriculture, horticulture, and forestry. Micropropagation is beneficial because by this technique, a large number of offspring or plantlets can be obtained in a short period of time.
- Production of disease-free plants: By meristem culture, disease free plants can be obtained.
- Embryo rescue: Interspecific hybris are often sterile because of embryo mortality and seed collapse. In such cases, the hybrid embryo is excised from the female parent in the early stage and is cultured in a culture medium. The interspecific hybrids may be several advantages over the original forms.
- Induction and selection of mutants: Using the tissue culture technique, mutation can be introduced in the cultures. The resistant mutants can be selected to produce resistant varieties.
- Somaclonal variations: Somaclonal variations are the differences in those plants that are raised from the plants by tissue culture. Such plants are tolerant to pests, disease or stresses.
- Genetic manipulation and somatic cell fusion: Genetic manipulation and somatic cell fusion in plant cells are possible in protoplast culture. But it is not possible in the plant cell because of its thick cellulose wall. It provides chance to get hybrid variety between non related species.
Micropropagation
Micropropagation is the process of producing thousands or millions of plants quickly by multiplying plant components in vitro under aseptic and regulated environmental conditions. Plant tissue culture is involved. A stock plant that does not generate seeds or does not react well to vegetative reproduction can also be utilized to produce a sufficient number of plantlets for planting.
Application of micropropagation
- In vitro propagation is a powerful and attractive tool for the rapid cloning of desirable plants. Newly selected cultivars can be bulked up rapidly to the level of several thousands in months rather than in years. Additionally, in tissue culture, plant multiplication can continue throughout the year irrespective of the season.
- Some plants, which do not multiply by seeds can be propagated through this plant tissue culture technique.
- Majority of vegetatively propagated crops are susceptible to virus infection. The technique of meristem tip culture of mother plant is now the best means of virus elimination from the infected stock.
- In some crops, the production of hybris seeds is very expensive. Production cost of such seeds may be reduced by in vitro multiplication of selected plants.
- Micropropagation has a great value as a potential system of germplasm storage.
- Tissue culture can be used to minimize the growing space in commercial nurseries for the maintenance of stock plants.